An extremely rare case of metastatic retinoblastoma of parotids presenting as a massive swelling in a child
Introduction
Retinoblastoma (Rb) is the most common ocular malignancy in childhood (1) with an annual incidence of 1 in 15,000 to 1 in 18,000 live births (2). Rb generally presents with leukocoria, diminution of vision, strabismus, proptosis or even developmental delay (1). Extraocular spread of Rb can be to brain and spine while it metastasizes to bone and bone marrow (1). However metastasis of Rb to the salivary glands is extremely rare with involvement of parotids have been mentioned by only 4 previously reported cases (3-5). Soni et al. (3) reported the first ever case of Rb metastasizing to parotids in 1978 and our case is the fifth such case till date. Similarly, metastatic spread to the parotids is also rare with cutaneous malignancies of the head and neck, renal cell carcinoma, carcinoma breast, small cell carcinoma lung, carcinoma stomach, splenic malignancy and rhabdomyosarcoma are some of the known primaries that have been reported (6). Patients of extraocular and metastatic Rb have had a poor prognosis (7) but recent approach of combining chemotherapy with radiotherapy has shown a better outcome (1) as is seen with our patient.
Case presentation
A 2-year-old male child presented with history of progressive enlargement of left eye associated with diminution of vision of 2 months duration. Clinical examination showed extensive proptosis of left eye with periorbital swelling. There was no leukocoria or strabismus in either eye. Left eye had no perception of light while right eye had a visual acuity of 6/12. Child underwent exenteration of left eyeball as MRI was suggestive of bilateral Rb with involvement of left optic nerve. Post-operative histopathological report (HPR) revealed poorly differentiated Rb. There was no history of Rb in his family. After 4 days the child developed swelling left parotid area, fine needle aspiration cytology (FNAC) of which showed small round blue cell tumor suggestive of metastasis to parotid. He received six cycles of chemotherapy inj. vincristine, etoposide and carboplatin (VEC).
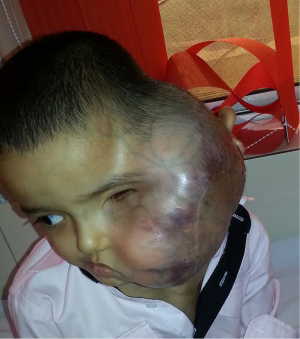
Post chemotherapy he lost to follow up, but after one year presented to us with a massive soft tissue swelling left side face associated with severe headache, nausea and vomiting. On examination the swelling measured 26 cm × 23 cm extending from occiput to left anterior chest wall (Figure 1). His mother’s history and treatment documents revealed that during the past 1 year he had two recurrences at the left parotid area for which he had undergone superficial parotidectomy twice in some other hospital where post-operative HPR showed malignant round cell tumor metastatic to parotid in a known case of Rb.
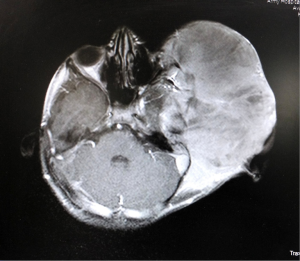
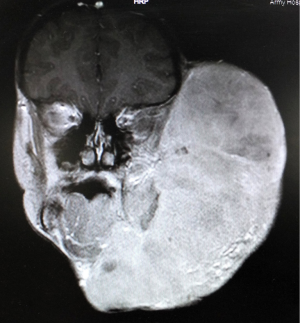
Magnetic resonance imaging (MRI) brain and face (Figure 2) at our institute showed a well-defined large lobulated mass lesion measuring 17.3 cm × 11.5 cm × 20 cm left side of face, neck, upper anterior chest, superiorly extending to left fronto-temporal scalp and inferiorly upto left anterior chest wall. It caused erosion of ramus of mandible, muscles of mastication, oral cavity, displacing trachea and esophagus towards right. The lesion was encasing the left common carotid artery. The lesion caused erosion of squamous temporal bone with intracranial extension to left temporal lobe (Figure 3).
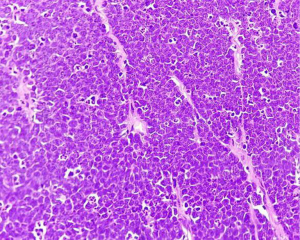
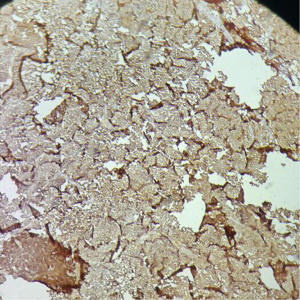
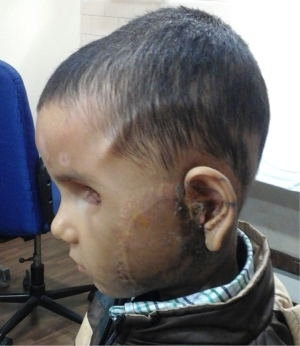
Biopsy from the left parotid swelling (Figure 4) showed malignant round blue cell tumor infiltrating parotids with IHC displaying immunopositivity for neuron specific enolase (NSE) (Figure 5) and immunonegativity for leukocyte common antigen (LCA), glial fibrillary acidic protein (GFAP) and vimentin consistent with Rb. Child was exhibited 3 cycles of alternating chemotherapy inj. vinblastin, doxorubicin, cyclophosphamide (VAC) and inj. ifosfamide and etoposide (IE) to which he responded well with reduction of swelling. After completion of chemotherapy he was treated with radiotherapy to face and brain to a dose of 40 Gray (Gy) in 20 fractions by 3 dimensional conformal radiotherapy (3DCRT) technique. The child tolerated RT well and showed significant response with symptomatic relief and complete resolution of left facial swelling (Figure 6). Presently the child is on regular follow-up without any evidence of recurrence or any new metastasis.
Discussion
Rb is the most common intraocular malignancy in children (1) representing less than 4% of all pediatric malignancies with an incidence of 1:15,000 to 1:18,000 live births (2). Rb affects approximately 250 to 300 children per year in the United States (1,2) while the average incidence rate in a developing country like India is 3.5 per million population (8). The average age of diagnosis of Rb is 18 months with 90% cases being diagnosed in children less than 5 years of age without any racial or sex predilection. The reason for higher incidence of Rb in developing countries as compared to developed countries is not known, however the presence of human papilloma virus and low socio-economic status is generally implicated (1).
In the developed world more than 95% of children suffering from Rb survive while only 50% children survive in developing nations (9). This trend can be due to poverty, low socio-economic status, illiteracy, lack of access to tertiary oncology centres thus causing a delay in presentation and, consequently a delay in diagnosis and subsequent treatment. Also a greater number of cases with extra-ocular extension and metastasis are seen in developing countries as compared to developed nations where the incidence is only 2% to 5% (7).
Rb most commonly presents with cat’s eye reflection or leukocoria (1) in the pupil. Other less common clinical signs and symptoms include diminution of vision, a red irritated uveitis-like eye, orbital cellulitis, phthisis bulbi, hyphema, buphthalmos, proptosis or a fungating mass as was seen in our patient. Rb spreads by direct invasion of optic nerve, via subarachnoid space to brain and spinal cord and generally metastasizes to bone and bone marrow (1) while salivary gland metastasis is uncommon. Patients may present with bone pains, nausea, vomiting and headache due to intracranial extension, growth retardation and anemia due to bone marrow invasion.
Rb can be unilateral or bilateral, heritable or non-heritable. Bilateral cases are mostly heritable though often without a family history (1) as was with our patient, but whether these factors determine metastasis to salivary glands is not clear. Soni et al. (3) in 1978 reported a unilateral Rb, Lussier et al. (4) in 2000 diagnosed one unilateral and one bilateral Rb, Khademi et al. (5) in 2003 reported a bilateral case and we in 2015 report this case of bilateral Rb, thus predicting a higher propensity of bilateral disease metastasizing to salivary glands or to parotids. Family history may be present in less than 10% of the cases (10).
Parotid gland malignancies are itself fairly uncommon accounting for only 3–6% of head and neck carcinomas and 0.3% of all malignancies (11). Metastatic spread to the major salivary glands may take place by lymphatic spread, hematogenous dissemination or by direct extension with parotids being the primary site of lympho-hematogenous spread as it contains around 20 to 30 lymph nodes and a dense network of lymphatic channels (12). Patients harboring metastatic lesion of parotids have an overall worse prognosis with the estimated 5-year survival rate being about 12.5% (12). The known prognostic factors for survival are the cell type, regional spread of the disease, primary malignancy and prevention of recurrence (12). Primary malignancies known for parotid metastasis are squamous cell carcinomas and melanomas of head and neck, renal cell carcinoma, carcinomas of lung, breast, gastro-intestinal tract, spleen and rhabdomyosarcoma (6).
A parotid swelling can be the initial presentation of any primary neoplasm and therefore should be evaluated upfront. A whole body PET scan serves as the primary modality for metastatic evaluation and knowing the location of the primary lesion but only a histopathological examination of the tissue can determine the definite diagnosis. Rb is characterized by the presence of small round cells with hyperchromatic nuclei, Flexner-Wintersteiner and Homer-Wright rosettes (1). Immunohistochemical staining is widely used to detect specific molecular markers characteristic of any particular cellular event, understand the distribution and localization of biomarkers and differentially expressed proteins in different parts of a biological tissue. In our case, IHC positivity for NSE while negativity for LCA, Desmin and Tdt was used to differentiate between Rb, Rhabdomyosarcoma and Ewing’s sarcoma, all belonging to the family of small round blue cell tumor and thus establishing the diagnosis of Rb metastasizing to parotid glands. NSE is specific for Rb cells and is found to be greater in bilateral cases (13).
Rb patients with extra-ocular or metastatic disease are known to have a poor prognosis, however recent studies using chemotherapy and RT have been quite encouraging (14). The use of chemoreduction with VAC or VEC followed by RT to a dose of 45 Gy to the involved sites yielded a 5-year event free or recurrence free survival rate of 84% in patients with orbital or preauricular disease (1,14). Chemoreduction before RT not only cause reduction in metastatic tumor bulk, controls micrometastasis and but also reduces the possibility of RT induced complications like growth deformities of skull and orbits, second malignancies like osteosarcoma, malignant melanoma, leukemia and cataract (1). The use of autologous stem cell rescue combined with high dose chemotherapeutic agents like carboplatin, etoposide, cyclophosphamide, melphalan and thiotepa with conformal RT has proved to improve survival up to 50% to 75% in patients with bulky metastatic disease without central nervous system involvement (15,16).
Metastasis to parotids is extremely rare, however the clinical outcome and overall prognosis of patients with parotid metastasis from Rb treated with multi-modality therapy is difficult to predict due to limited number of reported cases in literature. By reporting this case we recommend that the diagnosis of metastasis to parotids should be always be considered in a child with prior history of Rb presenting with a parotid gland mass and histopathology showing small round cell tumor positive for NSE, so as to enable a prompt initiation of appropriate therapy.
Acknowledgements
We thank the Next of Kin (NOK) of the child for allowing us to publish the case report and use the images taken while he was admitted in our institute.
We also like to extend our gratitude to department of Pediatrics, department of Pathology, department of Nuclear Medicine, and department of Radiology, Army Hospital Research and Referral, New Delhi, India.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent to publication was obtained from the NOK of the patient which has been submitted.
References
- Halperin EC, Brady LW, Perez CA, et al. Perez and Brady’s principles and practice of radiation oncology, sixth edition. Philadelphia: Lippincott Williams and Wilkins, Walters Kluwer Business, 2013;704-8.
- Abramson DH. Retinoblastoma incidence in the United States. Arch Ophthalmol 1990;108:1514. [Crossref] [PubMed]
- Soni NK, Arora HL, Chatterji P. Massive metastatic retinoblastoma of the parotid gland. J Laryngol Otol 1978;92:1049-52. [Crossref] [PubMed]
- Lussier C, Klijanienko J, Vielh P. Fine-needle aspiration of metastatic nonlymphomatous tumors to the major salivary glands: a clinicopathologic study of 40 cases cytologically diagnosed and histologically correlated. Cancer 2000;90:350-6. [Crossref] [PubMed]
- Khademi B, Derakhshandeh V, Vasei M, et al. Metastasis of retinoblastoma to the parotid gland: diagnosis by fine needle aspiration cytology. Otolaryngol Head Neck Surg 2003;128:296-7. [Crossref] [PubMed]
- Bron LP, Traynor SJ, McNeil EB, et al. Primary and metastatic cancer of the parotid: comparison of clinical behavior in 232 cases. Laryngoscope 2003;113:1070-5. [Crossref] [PubMed]
- Jubran RF, Erdreich-Epstein A, Butturini A, et al. Approaches to treatment for extraocular retinoblastoma: Children's Hospital Los Angeles experience. J Pediatr Hematol Oncol 2004;26:31-4. [Crossref] [PubMed]
- Yeole BB, Advani S. Retinoblastoma: An epidemiological appraisal with reference to a population in Mumbai, India. Asian Pac J Cancer Prev 2002;3:17-21. [PubMed]
- Leal-Leal C, Flores-Rojo M, Medina-Sansón A, et al. A multicentre report from the Mexican Retinoblastoma Group. Br J Ophthalmol 2004;88:1074-7. [Crossref] [PubMed]
- Meel R, Radhakrishnan V, Bakhshi S. Current therapy and recent advances in the management of retinoblastoma. Indian J Med Paediatr Oncol 2012;33:80-8. [Crossref] [PubMed]
- Spiro RH. Diagnosis and pitfalls in the treatment of parotid tumors. Semin Surg Oncol 1991;7:20-4. [Crossref] [PubMed]
- Ellis GL, Auclair PL, Gnepp DR. Surgical Pathology of the Salivary Glands. Philadelphia: WB Saunders; 1991.
- Kivelä T. Neuron-specific enolase in retinoblastoma. An immunohistochemical study. Acta Ophthalmol (Copenh) 1986;64:19-25. [Crossref] [PubMed]
- Chantada G, Leal-Leal C, Brisse H, et al. Is it pre-enucleation chemotherapy or delayed enucleation of severely involved eyes with intraocular retinoblastoma that risks extraocular dissemination and death? J Clin Oncol 2011;29:3333-4; author reply 3335-6. [Crossref] [PubMed]
- Kremens B, Wieland R, Reinhard H, et al. High-dose chemotherapy with autologous stem cell rescue in children with retinoblastoma. Bone Marrow Transplant 2003;31:281-4. [Crossref] [PubMed]
- Matsubara H, Makimoto A, Higa T, et al. A multidisciplinary treatment strategy that includes high-dose chemotherapy for metastatic retinoblastoma without CNS involvement. Bone Marrow Transplant 2005;35:763-6. [Crossref] [PubMed]