Treatment of Kawasaki disease by different doses of immunoglobulin: a meta analysis of efficacy and safety
Kawasaki disease (KD), an acute exanthematous disease characterized by systemic vasculitis, has shown an increasing trend of incidence over the past years (1). From the epidemiological viewpoint, it is known that this disease is more frequently found in Asiatic, and principally in Japanese, population. In North America, the highest number of cases are reported annually in the U.S. (2,3). The etiology includes very diverse bacterial and viral agents, chemical substances and acaros (dermatophagoides) without fully convincing evidence (4-6).
KD is a systemic vasculitis that occurs most commonly in children younger than 5 years of age (7). The inflammation involves small and medium-sized arteries, and the most severe problems arise from coronary artery inflammation. Coronary inflammation results in damage to the coronary arteries and may lead to artery aneurysms, arterial stenosis, and myocardial infarction (1,8). Such injuries may lead to serious problems in former patients during middle age. Research has shown that 15-26% of KD children will develop coronary artery aneurysms (9). Therefore, the aim of KD treatment is the prompt termination of systemic inflammation as early as possible and inhibition of the development of systemic vasculitis. Standard treatment for KD includes intravenous immunoglobulin (IVIG) and oral aspirin (ASA). Although the use of immunoglobulin has improved the prognosis for KD and dramatically lowered the incidences of cardiovascular diseases. However, some childern show persistent or recrudescent fever despite treatment with high-dose IVIG. And the doses of immunoglobulin and their efficacies and safety profiles remain controversial (10). Indicating that current alternative treatments are required for different patients.
Studies have shown that the dose of 2 g/(kg•d) for single use can effectively reduce the coronary artery lesions and shorten the fever disappearance time (11,12). However, other authors believed 2 g/(kg•d) for single use will result in the sudden increase of the plasma osmotic pressure of immunoglobulin, and thus destablize the homeostasis, increase the serum viscosity, and decrease blood flow velocity, which ultimately will facilitate the formation of blood clots and increase the incidence of stroke in the adulthood (13,14). In addition, some authors also believed that the dose of 1 g/(kg•d) for 1-2 days had equal efficacy as 2 g/(kg•d) for single use (15). Therefore, a comprehensive evaluation of different dosages of immunoglobulin in treating KD will be helpful to clarify their effectiveness and safety.
Subjects and methods
Inclusion and exclusion criteria of the studies
Randomized controlled trials, whether adopting blinding methods or allocation concealment, were enrolled in this analysis.
The inclusion criteria of subjects were as follows: (I) Pediatric patients confirmed to be with KD; and (II) patients who had received immunoglobulin therapy and at least undergone echocardiography within two weeks after treatment. Patients with other exanthematous diseases were excluded.
Interventions were compared as follows: Immunoglobulin 1 g/(kg•d) for 1-2 days vs. 2 g/(kg•d) for single use; immunoglobulin 1 g/(kg•d) for 1-2 days vs. 400 mg/(kg•d) for 4-5 days; and immunoglobulin 2 g/(kg•d) for single use vs. 400 mg/(kg•d) for 4-5 days.
The primary measures included: the incidences of coronary artery lesions (coronary artery dilatation and/or coronary aneurysm) in the acute phase (week 1-3), subacute phase (week 3-6), and follow-up (month 6 and 12 after treatment). The secondary indicators included adverse reactions (e.g., chills, rash, shock, irritability, and palpitation) and fever disappearance time.
Search strategy
Literature was searched using the pre-determined strategy based on thesaurus terms and free-text terms. Using the searching terms including gamma globulin, gamma-globulins, Y globulin, immunoglobulin, immunoglobulins, Kawasaki disease, mucocutaneous lymph node syndrome, and their Chinese counterparts, we electronically searched the databases including PubMed, EMBASE, Cochrane Library, CNKI, VIP and Wanfang.
Data extraction and quality evaluation
Two reviewers independently conducted searching based on the searching strategy and retrieved the data using a pre-determined data extraction form. The quality of the included studies was evaluated using the criteria (16) recommended by the Cochrane Handbook for Systematic Reviews of Interventions 5.0 in terms of randomization method, assignment concealment, blinding method, integration of result data, selective reporting of data and results; and other sources of bias. Disagreement between them was resolved through discussion or by consulting a third party.
Statistical analysis
Statistical analysis was performed using the Cochrane Collaboration’s software RevMan 5.0. Efficacy analysis was based on relative risk (RR) and weighted mean differences (MD). A 95% confidence interval was applied for each effect size. The heterogeneity among the enrolled studies were analyzed using chi square test, and a fixed effects model was applied when P>0.1 and I square (I2) <50%; If there was significant heterogeneity (P<0.1, I2>50%), the causes of such heterogeneity was searched, and subgroup analysis or sensitivity analysis was performed based on the clinical and methodological heterogeneity factors; if no clinical or methodological heterogeneity was found among the studies, random effect model was used.
Results
Search results
The initial search yielded 2,227 articles, which included 612 articles from PubMed, 90 from EMbase, 65 from Cochrane Library, 49 from VIP, 1,488 from CNKI, and 33 from Wangfang. Then, 110 duplicate reports were excluded. By reviewing the titles and abstracts, we ruled out 2,103 articles that did not meet the inclusion criteria. The full text of the remaining 124 potentially eligible articles were read; finally, 96 of them were excluded and 28 RCT (17-30) entered the final analysis (31-44).
Ten RCT (18,21,23,28-30,34,42-44) reported the use of 1 g/(kg•d) for 1-2 d vs. 2 g/(kg•d) for single sue, 16 (22,24,25,26-32,39-42) reported the use of 1 g/(kg•d) for 1-2 days vs. 400 mg/(kg•d) for 4-5 days, and 13 (17,19,20, 25,29,30,33,35,37,38,40,41,44) reported the use of 2 g/(kg•d) for single use vs. 400 mg/(kg•d) for 4-5 days. There were 3 studies (17,19,20) and 25 domestic studies (18,21-44) (Table 1).
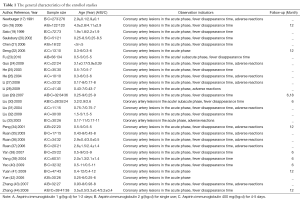
Full table
Results of quality assessment for the enrolled studies
All the enrolled 28 RCT mentioned “randomization”; however, only two studies (19,34) applied random digits table for randomization, two clearly (17,42) stated that they used sealed envelopes for concealed allocation, and two (17,42) used single-blind method (blind physician). Three studies (18,22,24) mentioned lost to follow-up, but failed to state whether intention-to-treat was applied (Table 2).
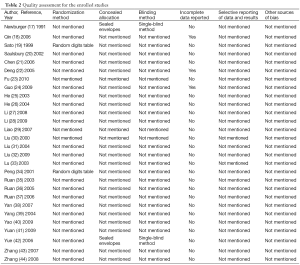
Full table
Comparison of the efficacy
Immunoglobulin 1 g/(kg•d) for 1-2 days vs. 2 g/(kg•d) for single use
Nine studies (19,21,23,29,30,34,42-44) reported the incidence of coronary artery injuries at the acute phase and two (23,30) reported the incidence of coronary artery injuries at the sub-acute phase; Meta analysis showed that these two dosages showed no significant difference in terms of the incidences of coronary artery injuries at the acute and sub-acute phases (P>0.05). Two studies (29,30) reported the incidence of coronary artery injuries at the 6-month follow-up and four (18,29,34,44) reported that at 1-year follow-up; Meta analysis showed that these two dosages showed no significant difference in terms of the incidences of coronary artery injuries at 6-month and 1-year follow-up (P>0.05). Four studies (18,29,34,44) reported the adverse reactions of immunoglobulin; analysis showed that the incidences of adverse reactions showed no significant differences between these two groups (P>0.05). Five studies (18,21,30,34,44) reported the fever disappearance time after the use of immunoglobulin; analysis showed that the fever disappearance time was not significantly different between these two groups (P>0.05) (Table 3).

Full table
Immunoglobulin 1 g/(kg•d) for 1-2 days vs. 400 mg/(kg•d) for 4-5 days
Eleven studies (22,24,26,28-32,36,37,42) reported the incidence of coronary artery injuries at the acute phase and one study (25) reported the incidence of coronary artery injuries at the sub-acute phase; Meta analysis showed that these two dosages showed no significant difference in terms of the incidences of coronary artery injuries at the acute and sub-acute phases (P>0.05). Four studies (28-30,39) reported the incidence of coronary artery injuries at the 6-month follow-up and three studies (22,24,44) reported the incidence of coronary artery injuries at the 1-year follow-up; Meta analysis showed that these two dosages showed no significant difference in terms of the incidences of coronary artery injuries at 6-month follow-up (P>0.05); One study (44) showed that the incidence of coronary artery injuries at 1-month follow-up was not significantly different between these two groups (P>0.05). Seven studies (27-29,32,36,39,44) reported adverse reactions. The incidences of adverse reactions showed no significant differences between these two groups (P>0.05). Ten studies (22,24,25,27,28,30-32,36,44) reported the fever disappearance time after the use of immunoglobulin; Meta analysis showed that the fever disappearance time was significantly shorter in the 1 g/(kg•d) for 1-2 days group than in 400 mg/(kg•d) for 4-5 days group (P<0.001) (Table 4).
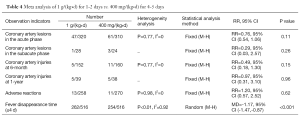
Full table
Immunoglobulin 2 g/(kg•d) for single use vs. 400 mg/(kg•d) for 4-5 days
Nine studies (17,19,26,29,30,33,35,37,44) reported the incidence of coronary artery injuries at the acute phase and one study (30) reported the incidence of coronary artery injuries at the sub-acute phase. Meta analysis showed that the incidence of coronary artery injuries at the acute phase was significantly lower in the 2 g/(kg•d) for single use group than in 400 mg/(kg•d) for 4-5 days group (P<0.05), although that at the sub-acute phase showed no significant difference between these two groups (P>0.05). Four studies (29,30,38,40) and one study (44), respectively, reported the incidence of coronary artery injuries at the 6- and 12-month follow-up. Meta analysis showed that the incidence of coronary artery injuries was significantly lower in the 2 g/(kg•d) for single use group than in 400 mg/(kg•d) for 4-5 days group at the 6-month follow-up (P=0.009), although it showed no significant different at 12-month follow-up (P=0.90). Five studies (17,19,26,29,44) reported the adverse reactions of immunoglobulin; the incidences of adverse reactions showed no significant differences between these two groups (P>0.48). Eleven studies (17,19,20,25,33, 35,37,38,40,41,44) reported the fever disappearance time after the use of immunoglobulin; Meta analysis showed that the fever disappearance time was significantly shorter in the 2 g/(kg•d) for single use group than in 400 mg/(kg•d) for 4-5 days group (P<0.001) (Table 5).
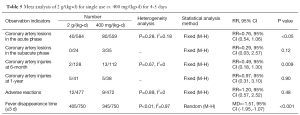
Full table
Discussion
Our analysis found that, compared with the immunoglobulin 2 g/(kg•d) for single use, immunoglobulin 1 g/(kg•d) for 1-2 days showed no significant differences in terms of the incidences of coronary artery injuries at the acute phase, sub-acute phase, 6-month follow-up, and 12-month follow-up, the fever disappearance time, and the incidences of adverse reactions. Although a study enrolling 1,052 pediatric patients indicated that the incidence of coronary artery injuries at the sub-acute phase was lower in the 2 g/(kg•d) for single use group (13), the study was a retrospective case-control study, which is inferior to RCT in controlling the selection bias, performance bias, and attrition bias. Our analysis also found that, compared with the immunoglobulin 400 mg/(kg•d) for 4-5 days, the immunoglobulin 1 g/(kg•d) for 1-2 days had significantly shorter fever disappearance time, although it showed no significant difference in terms of the incidences of coronary artery injuries at the acute phase, sub-acute phase, 6-month follow-up, and 12-month follow-up and the incidences of adverse reactions. Furthermore, although the 2 g/(kg•d) for single use group was non-superior to 400 mg/(kg•d) for 4-5 days group in terms of the incidences of coronary artery injuries at the sub-acute phase and 6-month follow-up and the incidences of adverse reactions, it showed certain advantages in the terms of the incidence of coronary artery injuries at the acute phase and 6-month follow-up and the fever disappearance time. Mofi et al. (45) found the 2 g/(kg•d) for single use had better efficacy, shorter fever disappearance time, and lower incidence of coronary artery injuries. In addition, our pooled analysis showed the fever disappearance time was highly heterogeneous (P<0.1, I2>50%), which may be explained by the different physical status and drug sensitivities of the individual patient.
Among the 28 enrolled RCT, only two described the correct randomization methods and two used single-blind method. Therefore, there was unavoidable case-selection bias.
Immunoglobulin can cause mild adverse reactions such as chills, rash, shock, irritability, and palpitation, and also may induce severe adverse reactions such as embolism and myocardial infarction (13,14). All the enrolled literature in this analysis mentioned mild adverse reactions; however, no literature mentioned these severe adverse reactions or the increased risk of stroke during adulthood due to the short follow-up period. For the long-term efficacy, only 6 studies mentioned the outcomes of coronary artery injuries that had been followed up for one year; in other words, few long-term follow-up has been conducted for these injuries. In their studies, Deng et al. (22) and Guo et al. (24) mentioned that some patients were lost during the 1-year follow-up; however, they did not report the numbers of patients lost to follow-up in the treatment group and control group separately, making it impossible to merge the data. Therefore, standardized data reporting should be emphasized in the future studies.
In summary, There are similar therapeutic effects for KD between the immunoglobulin treatment groups at the doses of 1 g/(kg•d) for 1-2 days and 2 g/(kg•d) for single use. All the indicaters show no remarkable difference between these two doses except that the dose of 2 g/(kg•d) for single use is superior to 400 mg/(kg•d) for 4-5 days in terms of the incidences of coronary artery injuries at the acute phase and 6-month follow-up while the dose of 1 g/(kg•d) for 1-2 days has shorter fever disappearance time than 400 mg/(kg•d) for 4-5 days. However, the long-term efficacy (especially for coronary artery abnormalities) and safety of immunoglobulin still require further investigation.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Kim DS. Kawasaki disease. Yonsei Med J 2006;47:759-72. [PubMed]
- Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 2004;110:2747-71. [PubMed]
- Burns JC, Glodé MP. Kawasaki syndrome. Lancet 2004;364:533-44. [PubMed]
- Zhi CL, Migita M, Hayakawa J, et al. Establishment of modified retroviral vector targeting X-linked severe combined immunodeficiency. J Nippon Med Sch 2004;71:51-6. [PubMed]
- Falcini F. Kawasaki disease. Curr Opin Rheumatol 2006;18:33-8. [PubMed]
- Sotelo N, González LA. Kawasaki disease: a rare pediatric pathology in Mexico. Twenty cases report from the Hospital Infantil del Estado de Sonora. Arch Cardiol Mex 2007;77:299-307. [PubMed]
- Tse SM, Silverman ED, McCrindle BW, et al. Early treatment with intravenous immunoglobulin in patients with Kawasaki disease. J Pediatr 2002;140:450-5. [PubMed]
- Klassen TP, Rowe PC, Gafni A. Economic evaluation of intravenous immune globulin therapy for Kawasaki syndrome. J Pediatr 1993;122:538-42. [PubMed]
- Cha S, Yoon M, Ahn Y, et al. Risk factors for failure of initial intravenous immunoglobulin treatment in Kawasaki disease. J Korean Med Sci 2008;23:718-22. [PubMed]
- Liang CM, Lee PC, Hwang BT, et al. Comparison of different types and regimens of intravenous immune globulin (IVIG) in patients with Kawasaki disease. Acta Cardiol Sin 2004;20:15-20.
- Rigante D, Valentini P, Rizzo D, et al. Responsiveness to intravenous immunoglobulins and occurrence of coronary artery abnormalities in a single-center cohort of Italian patients with Kawasaki syndrome. Rheumatol Int 2010;30:841-6. [PubMed]
- Sakata K, Hamaoka K, Ozawa S, et al. A randomized prospective study on the use of 2 g-IVIG or 1 g-IVIG as therapy for Kawasaki disease. Eur J Pediatr 2007;166:565-71. [PubMed]
- Du ZD, Zhao D, Du JB, et al. Effects of three intravenous gamma globulin regimens on Kawasaki disease and relevant coronary complication rates in pediatric patients: a comparative study. Zhonghua Yi Xue Za Zhi 2007;87:3119-21. [PubMed]
- Baba R. Effect of immunoglobulin therapy on blood viscosity and potential concerns of thromboembolism, especially in patients with acute Kawasaki disease. Recent Pat Cardiovasc Drug Discov 2008;3:141-4. [PubMed]
- Shulman ST. Kawasaki disease and IVIG: what’s going on here? Immunotherapy with intravenous immunoglobulins. San Diego: Academic Press 1991:261-8.
- Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.0 [updated February 2008] [DB/OL]. The Cochrane Collaboration 2008. Available online: www.Cochrane-handbook.org
- Newburger JW, Takahashi M, Beiser AS, et al. A single intravenous infusion of gamma globulin as compared with four infusions in the treatment of acute Kawasaki syndrome. N Engl J Med 1991;324:1633-9. [PubMed]
- Qin LJ, Wang HW, Hu XF, et al. Therapeutic effectiveness of intravenous immunoglobulin at 1 g/kg and 2 g/kg on Kawasaki disease: a comparative and follow-up study. Zhonghua Er Ke Za Zhi 2006;44:891-5. [PubMed]
- Sato N, Sugimura T, Akagi T, et al. Selective high dose gamma-globulin treatment in Kawasaki disease: assessment of clinical aspects and cost effectiveness. Pediatr Int 1999;41:1-7. [PubMed]
- Saulsbury FT. Comparison of high-dose and low-dose aspirin plus intravenous immunoglobulin in the treatment of Kawasaki syndrome. Clin Pediatr (Phila) 2002;41:597-601. [PubMed]
- Chen BQ, Shi WZ. Therapeutic effects of medium-dose gamma globulin on Kawasaki disease. Zhejiang Clinical Medical Journal 2006;8:308.
- Deng MY. Comparison of two gamma globulin regimens in the treatment of Kawasaki disease. International Medicine & Health Guidance News 2005;11:33-4.
- Fu JN, Chen HP. Comparison of different dose intravenous gamma-globulin in the treatment of Kawasaki disease. Chinese Journal of Modern Drug Application 2010;4:121-2.
- Guo L, Zhang HB, Dou JL. Curative effect observation and nursing experience of different dosage of intravenous immunoglobulin on the treatment of Kawasaki disease. China Clin Prac Med 2009;3:116-7.
- Zhang Q, Li JY, He XX. Therapeutic effects of different dose of intravenous immuneglobulins in 65 patients with Kawasaki disease. Fujian Medical Journal 2003;25:49-50.
- He Z. Comparison of two intravenous gamma globulin regimens in the treatment of acute Kawasaki disease. Hebei Medical Journal 2004;26:323-3.
- Liao Y, Li XL. Curative effect of intravenous high dose gamma immunoglobulin in 33 children with Kawasaki disease. Journal of Gannan Medical University 2008;28:36-7.
- Li JG. Curative effect of two intravenous gamma globulin regimens on the treatment of Kawasaki disease. Practical Clinical Medicine 2009;10:60-1.
- Liao J, He KJ. The Clinical Study of Different Doseage Immune Globulin in Treating Kawasaki in Intravenous Injection. China Healthcare Innovation 2007;2:8-9.
- Liu XM. Dosage of intravenous immune globulin therapy for Kawasaki disease. Shandong Medical Journal 2000;40:23-4.
- Liu TJ. Therapeutic effects of intravenous immune globulin on Kawasaki disease. Modern Journal of Integrated Traditional Chinese and Western Medicine 2004;13:451.
- Liu M, Cheng J, Luo Q. Comparison of two intravenous gamma globulin regimens in the treatment of children with Kawasaki disease. Chinese Journal of Birth Health & Heredity 2009;17:117-8.
- Zheng ZL, Lu JX. Therapeutic effects of intravenous immune globulin on Kawasaki disease. Qinghai Medical Journal 2003;33:16-8.
- Peng CM, Xiao Q, Cheng YL, et al. A Study on Different Dose of Intravenous Immuneglobulins for Treating Kawasaki Disease. Journal of Nanhua University 2001;29:581-3. (Medical Edition).
- Ruan J, Cai SJ, Lin RZ, et al. Comparison of two gamma globulin regimens in the treatment of Kawasaki disease. Qingdao Medical Journal 2006;38:175-6.
- Ruan J, Yang HB, Ruan J, et al. Comparison of two high-dose gamma globulin in the treatment of Kawasaki disease. Practical Pharmacy and Clinical Remedies 2005;8:48-9.
- Ruan XL, Xiang RL, Liang GA, et al. Effects of different dose IVIG on CD4+/CD8+ of children with Kawasaki disease. Journal of Pediatric Pharmacy 2003;9:18-9.
- Yan P. Curative effect of different dosage of intravenous gamma globulin on the treatment of Kawasaki disease. Journal of Community Medicine 2007;5:11.
- Yang HX, Zheng MC. Comparison of two gamma-globulin regimens in the treatment of Kawasaki disease. Chin J Contemp Pediatr 2004;6:57-8.
- Yao XH. Treatment with different dosage of intravenous gamma globulin in 64 patients with Kawasaki disease. Chinese Community Doctors 2009;11:68-9.
- Kuai WX, Yuan YF. Role of gamma globulin in pediatric patients with Kawasaki disease. Shanxi Medical Journal 2009;38:365-6.
- Yue AH, Tian QL, Li H. Curative effect of intravenous gamma globulin in 56 patients with Kawasaki disease. Clinical Focus 2006;21:962-3.
- Zhang YQ. Curative effect of two high-dose intravenous gamma globulin on the treatment of Kawasaki disease. Sichuan Medical Journal 2007;28:801-2.
- Zhang WH. A analysis on different dose of gamma globulin for treating Kawasaki disease. Journal of Practical Training of Medicine 2008;36:117-20.
- Mori M, Miyamae T, Imagawa T, et al. Meta-analysis of the results of intravenous gamma globulin treatment of coronary artery lesions in Kawasaki disease. Mod Rheumatol 2004;14:361-6. [PubMed]


