
Evaluation of assessment of caregiver experience with neuromuscular disease: reliability and responsiveness of a new caregiver-reported outcome measure in patients with cerebral palsy
Introduction
Cerebral palsy (CP) is the most common cause of chronic childhood disability and frequently occurs with motor, sensory and/or cognitive deficits. For children with CP and limited capacity for self-care, parents must often provide extensive, prolonged care throughout most of their children’s lives (1-4). These children commonly also have comorbid neurological disorders such as epilepsy or other medical conditions that contribute to poor patient health and greater dependency needs (1). While the underlying illness causing CP may be progressive or not, worsening neuromuscular function tends to occur even in patients with static central nervous system lesions (5).
Appreciation for what is termed “caregiver impact” has grown generally in recent decades. Caregiver impact is a complex, multidimensional construct that involves the subjective perception of excessive strain in physical, psychological, emotional, social and/or financial domains, among others. As a subjective and internal process, the perception of impact varies among caregivers and may be diminished or exacerbated by caregiver-specific factors, such as psychological make-up and coping strategies, patient-specific factors, like temperament, and outside factors, including the presence or absence of familial, social and economic support structures. When sufficiently great, caregiver impact may negatively affect caregiver physical and psychological health (2,6-13). Among patients with CP, the tremendous impact on caregivers can lead to adverse psychological and physical health of both caregivers and patients.
Assessment of Caregiver Experience with Neuromuscular Disease (ACEND) (1), a caregiver impact-based reported outcome measure that has been validated in caregivers of children with CP, may represent a useful assessment tool to gauge the burden of care on caregiver impact and their perceived health-related quality of life (HRQoL) (2,6-13). Designed with the aim of assessing the caregiver impact experienced by parents of multi-handicapped children receiving orthopaedic intervention, ACEND contains 41 items which reflect 2 domains, including 7 sub-domains. Domain 1, examining physical impact, includes 4 sub-domains: feeding/grooming/dressing (6 items), sitting/play (5 items), transfers (5 items) and mobility (7 items). Domain 2, which examines general caregiver impact, includes 3 sub-domains: time (4 items), emotion (9 items), and finance (5 items). Score for each item is generated based on the 6- and 5-point ordinal scales and is such designed that caregivers experiencing less intense care-giving impact received higher scores. Sub-domain and total scores are thus meant to confer “impact health” in each caregiver respondent.
Spasticity is a very common finding among these children with CP and significant spasticity can lead to difficulty of care-giving, as well as pain and decreased function of the patient. Treatment of spasticity with botulinum toxin was first reported in 1993. Botulinum toxin provides a focal, controlled muscle weakness that leads to a reduction in unwanted spasticity. The current study aims to present the first evaluation of responsiveness of the ACEND as a measurement tool of caregiver impact and their perceived HRQoL among parents of children and adolescents with CP following botulinum toxin injection to relieve spasticity. We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/tp-19-176).
Methods
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by institutional review board of Columbia University (NO. AAAN6260) and informed consent was taken from all the patients. Patients (0–19 years of age) who presented at a tertiary pediatric orthopaedic center, who fit the inclusion criteria, were recruited into the study from 2012 to 2014. Patients carrying an ICD-10 diagnosis code of CP who required botulinum toxin treatment to decrease muscle spasticity in the lower extremities were eligible for this study. All patients received only one injection during the study period. Patients were recruited at any time point in treatment. Patients with non-English speaking parents were excluded from the study because the ACEND questionnaire used in the study protocol is only validated in English. The dataset contains demographic information (gender, age at procedure), diagnosis (ICD-10 Diagnosis Code), functional level (The Gross Motor Function Classification System (GMFCS), a 5-level clinical classification system that describes the gross motor function of people with CP on the basis of self-initiated movement abilities), and ACEND questionnaire-related information (baseline ACEND scores and ACEND scores at follow-up). The software package IBM SPSS Statistics 21.0 was used to compute the correlation between ACEND scores at baseline and follow-up for each study participant. Additionally, paired-sample t-test was used to further examine the reliability of ACEND. Lastly, linear regressions models were built for baseline, follow-up, and change in ACEND scores for study participants. Statistically significant predictors were identified. Akaike information criterion (AIC) is a measure of the relative quality of a statistical model for a given dataset and was used for the selection of the best linear regression model for ACEND score changes.
Results
A total of 41 patients (17 female and 24 male) who had both baseline ACEND scores and at least one score during follow-up were identified and enrolled into the study. Patient age at the time of botulinum toxin injection ranged from 2 to 20 years old. The average age of the 41 patients enrolled in the study was 9.6±5.4 years old. Follow-up time after botulinum toxin injection ranged from 1 month to 20 months. The average follow-up time was 6.4±4.7 months. Thirty-seven patients (90.2%) responded to only one ACEND assessment and only 4 patients (9.8%) responded to more than one assessment (as a result of their participating in other studies). Since the average follow-up was 6.4 months, ACEND scores collected at the time point closest to 6 months were used in this study for the 4 patients with multiple assessments. Five ICD-10 diagnosis codes were identified among the 41 study participants (Table 1): 9 were G80.1 (Congenital diplegia), 2 were G80.2 (Congenital hemiplegia), 7 were G80.0 (Congenital quadriplegia), 4 were G81.9 (Infantile hemiplegia), and 16 were G80.9 (Infantile CP). GMFCS levels for the 41 patients in the current study were determined by consensus by the treating orthopaedic surgeon and physical therapist. 10 study participants were GMFCS I, 5 were GMFCS II, 6 were GMFCS III, 9 were GMFCS IV, and 11 were GMFCS V (Table 2). The average baseline ACEND score was 117.7±47.7 and the scores at follow-up averaged at 120.4±49.5.
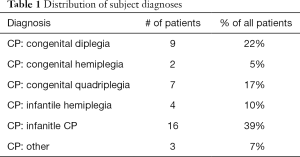
Full table
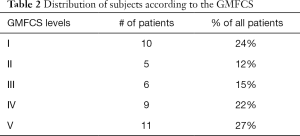
Full table
The baseline and follow-up ACEND scores for each study participants were found to be strongly correlated with a coefficient of 0.929 (P<0.001). Under paired-sample t-test, the average increase in ACEND score was found to be 2.7 (P=0.352). The strong correlation between the two scores for each study participant suggest that the ACEND questionnaire has high reliability among the population under study. Additionally, the effects of botulinum toxin should have largely worn off by six months, so not finding a significant change in the ACEND score is not necessarily an indicator of its ineffectiveness. Since botulinum toxin injection is a minimally invasive procedure, we did not anticipate a significant change in caregiver burden as reflected by ACEND. Furthermore, the slight increase of ACEND score is in line with our previous observation that caregiver burden typically increases and remain higher after a procedure for a relatively short period of time (median follow up time in this study was 6 months), even though the benefits of botulinum toxin in decreasing spasticity is well-known.
Linear regression for baseline ACEND score using ICD-10 diagnosis code, age at treatment, GMFCS level, and gender as predictors was conducted. F statistic of the model was 10.192 (P<0.001), suggesting that the model is valid for prediction of baseline ACEND scores. Two predictors, ICD-10 code and GMFCS level, were found to be statistically significant (P=0.043, P<0.001), suggesting that the pathology of the disease and the ambulatory function level of the patient were most important in predicting ACEND score at baseline.
Linear regression for ACEND score at follow-up including all predictors (ICD-10 diagnosis code, age at treatment, follow-up time, GMFCS level, and gender) was also conducted. F statistic of the model was 10.671 (P<0.001), suggesting that the model is valid for predicting baseline ACEND scores. Two predictors, ICD-10 diagnosis and GMFCS level, were found to be statistically significant (P=0.025, P<0.001), suggesting that the pathology of the disease and the ambulatory function level of the patient at baseline were most important in predicting ACEND score at follow-up, as is the case with baseline ACEND score prediction.
Additionally, a series of linear regression models for change in ACEND scores at follow-up from baseline were evaluated by first including all predictors, then removing the variable with the highest P value in a stepwise fashion, as long as the AIC of the model keeps decreasing, until the remaining predictors were all statistically significant at a type I error threshold of 5%. The final model had an F statistic of 9.814 (P<0.001) and included two predictors, gender and follow-up time. The coefficient for gender was 19.611, indicating that on average boys had an ACEND score change 19.611 points higher than that of girls.
Discussion
The care of children with CP can have a profound impact on the use of caregiver’s time and psychosocial health, a finding that has been documented in other areas of health care. The purpose of this project was to evaluate the reliability and responsiveness of a caregiver impact-based measure, the ACEND, and to identify the most important predictors for ACEND scores and their changes, in the population of children with CP.
The original validation found ACEND as a valid and disease-specific assessment tool to quantify the HRQoL of caregivers of children with CP (5). Since then, several studies have examined the use of ACEND in different clinical settings. Difazio et al. evaluated changes in the impact on caregivers and their perceived HRQoL after orthopaedic surgical correction of hip or spinal deformities among children with CP but reported no changes in any of the ACEND domains (14). A subsequent prospective longitudinal study by the same authors found that HRQoL of caregivers improved 1 year after spinal fusion but regressed to baseline after 2 years, while the burden of care remained the same, according to the ACEND scale (15). Another study seeking to identify associated factors for higher levels of CP caregiver stress using the ACEND scale demonstrated that worrying about the child’s pain and the financial concerns over lost wages were most highly reported areas of stress (16). Similarly, Vessey et al. found that the subscale scores as measured by ACEND indicated that children with CP who required hip or spine surgery had a significant impact on family finances (17).
This study suggested the reliability of ACEND through statistically significant, strong correlations in ACEND scores before and after botulinum toxin injection, a minimally-invasive clinical procedure used to control spasticity commonly seen in patients with CP. A slight increase in ACEND score was observed following botulinum toxin injection, in line with our anticipation that caregiver burden will increase for a short period of time after the procedure. Linear regression models on baseline ACEND scores, ACEND scores at follow-up, and the change between the two suggest that while disease pathology (ICD-10 diagnosis codes) and ambulatory function level (GMFCS) are the most important predictors for ACEND scores at both baseline and follow-up (further corroborating the reliability of the ACEND questionnaire), they are not statistically significant for prediction of ACEND score changes. Variables most important for prediction of such changes are gender and follow-up time. In other words, while the burden of care is largely determined by ICD-10 diagnosis and the GMFCS level, changes in the burden of care (as measured by ACEND questionnaire) are only related to the gender of the patient and the follow-up time interval. Our finding that boys on average demonstrated a greater increase in the ACEND score in the first several months following botulinum toxin injection suggests that the increase in burden of care was higher for caregivers of boys than for those of girls following this procedure. And lastly, the ACEND measure did not seem to be sensitive to the effects of botulinum toxin injection in our cohort of caregiver respondents (half of their children were GMFCS IV and V), and more studies with larger sample sizes and different patterns of GMFCS levels are needed to further validate the sensitivity of the ACEND measure after botulinum toxin injection among caregivers of children with CP.
Conclusions
This study was limited by its small size, inclusion of a relatively even distribution of patient characteristics, and the relatively short period of follow-up. Future studies, which are already underway, will include more patients and examine the subdomains of ACEND individually, and will focus on comparison across GMFCS levels and ICD-10 diagnoses to define functional level and disease pathology-specific caregiver impact profiles. Furthermore, similar studies will be conducted for more invasive surgical procedures, such as spinal instrumentation and hip osteotomies, which may have a more significant impact on caregiver burden postoperatively, leading to a more significant change in ACEND scores.
Acknowledgments
Funding: This is study was supported by a grant from Capital’s Fund for Health Development and Research (Grant No. 2018-4-4097). The organizations that funded this study had no role in the study design, data collection and analysis or the decision to prepare or publish the manuscript.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/tp-19-176
Data Sharing Statement: Available at http://dx.doi.org/10.21037/tp-19-176
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tp-19-176). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by institutional review board of Columbia University (No. AAAN6260) and informed consent was taken from all the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Green LB, Hurvitz EA. Cerebral palsy. Phys Med Rehabil Clin N Am 2007;18:859-82, vii. [Crossref] [PubMed]
- Hallum A, Krumboltz JD. Parents caring for young adults with severe physical disabilities: psychological issues. Dev Med Child Neurol 1993;35:24-32. [Crossref] [PubMed]
- Rang M, Wright J. What have 30 years of medical progress done for cerebral palsy? Clin Orthop Relat Res 1989.55-60. [PubMed]
- Stevenson CJ, Pharoah POD, Stevenson R. Cerebral palsy - The transition from youth to adulthood. Dev Med Child Neurol 1997;39:336-42. [Crossref] [PubMed]
- Matsumoto H, Clayton-Krasinski DA, Klinge SA, et al. Development and initial validation of the assessment of caregiver experience with neuromuscular disease. J Pediatr Orthop 2011;31:284-92. [Crossref] [PubMed]
- Boyer F, Drame M, Morrone I, et al. Factors relating to carer burden for families of persons with muscular dystrophy. J Rehabil Med 2006;38:309-15. [Crossref] [PubMed]
- Brehaut JC, Kohen DE, Raina P, et al. The health of primary caregivers of children with cerebral palsy: how does it compare with that of other Canadian caregivers? Pediatrics 2004;114:e182-91. [Crossref] [PubMed]
- Breslau N, Staruch KS, Mortimer EA Jr. Psychological distress in mothers of disabled children. Am J Dis Child 1982;136:682-6. [PubMed]
- Chou KR. Caregiver burden: a concept analysis. J Pediatr Nurs 2000;15:398-407. [Crossref] [PubMed]
- Grad J, Sainsbury P. Problems of caring for the mentally ill at home. Proc R Soc Med 1966;59:20-3. [Crossref] [PubMed]
- Hamzat TK, Mordi EL. Impact of caring for children with cerebral palsy on the general health of their caregivers in an African community. Int J Rehabil Res 2007;30:191-4. [Crossref] [PubMed]
- Raina P, O'Donnell M, Rosenbaum P, et al. The health and well-being of caregivers of children with cerebral palsy. Pediatrics 2005;115:e626-36. [Crossref] [PubMed]
- Raina P, O'Donnell M, Schwellnus H, et al. Caregiving process and caregiver burden: Conceptual models to guide research and practice. BMC Pediatr 2004;4:1. [Crossref] [PubMed]
- Difazio RL, Vessey JA, Zurakowski D, et al. Differences in health-related quality of life and caregiver burden after hip and spine surgery in non-ambulatory children with severe cerebral palsy. Dev Med Child Neurol 2016;58:298-305. [Crossref] [PubMed]
- DiFazio RL, Miller PE, Vessey JA, et al. Health-Related Quality of Life and Care Giver Burden Following Spinal Fusion in Children With Cerebral Palsy. Spine (Phila Pa 1976) 2017;42:E733-9. [Crossref] [PubMed]
- Lowes L, Clark TS, Noritz G. Factors associated with caregiver experience in families with a child with cerebral palsy. J Pediatr Rehabil Med 2016;9:65-72. [Crossref] [PubMed]
- Vessey JA, DiFazio RL, Strout TD, et al. Impact of Non-medical Out-of-pocket Expenses on Families of Children With Cerebral Palsy Following Orthopaedic Surgery. J Pediatr Nurs 2017;37:101-7. [Crossref] [PubMed]


