Wilms tumor with intravascular tumor thrombus
Wilms tumor (WT), also known as nephroblastoma, is the most common renal tumor in children. It accounts for more than 90% of all renal tumors in children.
The survival rates have greatly improved over the years, based on the currently available treatment protocols. Most of the patients with WT can expect to be cured of their disease following multimodal therapy (1,2).
Multicentre trials have been carried out both in Europe by Society of International Pediatric Oncology (SIOP) and in the United States by National Tumor Wilms Study Group (NTWSG) since 1980’s. By using chemotherapy and radiotherapy, in addition to total nephrectomy, the prognosis of WT has improved dramatically to more than 90% in recent years. The management of WT differs in Europe and North America. In Europe, WTs are treated according to the SIOP protocols. Use of preoperative chemotherapy provides tumor reduction prior to surgical resection in this approach. On the other hand, in North America, management of WT is according to the NTWSG regimens which advocate surgery prior to chemotherapy. Inoperable WT constitutes about 28% of total cases of WT. Large size of tumor, involvement of adjacent vital structures, and intracaval tumor thrombus are mostly accepted as inoperable criteria.
Overall survival and relapse free survival rates are almost same from different treatment strategies with both the NTWSG and SIOP approach.
WT has a strong tendency to invade blood vessels in the form of tumor thrombus, into the renal veins, and inferior vena cava and even into the right atrium. WT can extend into its venous drainage resulting in thrombus. Intravascular extension of WT to the renal vein has been reported to occur in up to 20% to 35% of patients (3-7). Extension of tumor thrombus along to the renal vein into the inferior vena cava occurs in 4-10% of all patients. Tumor thrombus extending to the right atrium is less reported as 0.7-1% (8-11) WT with renal vein thrombus has been reported to be more common (59-85%) in the right kidney because of the shorter right renal vein (Table 1).
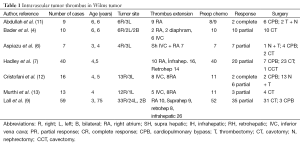
Full table
Most patients with tumor thrombus are asymptomatic and diagnosis is only made on imaging investigations. Ritchey et al. in a retrospective review of 77 patients with intravascular extension, observed only five with clinical findings suggesting this condition (12). Preoperative identification of caval thrombus in patients with retroperitoneal tumors is very important for surgery. The accuracy of various imaging modalities in detection of tumor thrombus in children with WT has not been defined. Several imaging modalities including computed tomography (CT), magnetic resonance imaging (MRI) and Doppler ultrasonography (USG) can demonstrate intravascular tumor thrombus before surgery (13,14). In addition to CT and MRI, Doppler USG is reliable in demonstrating the presence and extent of inferior vena cava tumor thrombus.
The current recommendation is that USG with color Doppler sonography should be performed for preoperative evaluation of tumor thrombus in WT. In patients with a suspected renal tumor, Doppler USG should be performed to detect tumor infiltration of renal vein and inferior vena cava and to assess patency of blood flow. Hadley et al. reported that they have found CT combined with Doppler USG to be useful in defining the presence and extent of tumor thrombus (7). But, on the other hand, some authors recently reported that CT can accurately identify cavoatrial tumor thrombus in their studies. McDonald et al. investigated the role of CT or MRI of abdomen over USG in children with WT. They concluded that CT or MRI added additional information in local staging of the patients (13). An echocardiogram will also add to the information on the extent of the intracardiac thrombus.
The classification of intracaval tumor thrombi is performed according to the upper extent of the tumor thrombus: level 1, infrahepatic thrombus; level 2, retrohepatic thrombus; level 3, suprahepatic thrombus; and level 4, atrial thrombus. The presence of cavoatrial tumor thrombus can lead to differences in management. The management of WT with tumor thrombus is determined by multiple factors such as extent of tumor thrombus, chemotherapy response of the tumor (15-18).
In traditional concept, the recommended treatment of WT with tumor thrombus extending into the renal vein is open surgery to avoid tumor spread and minimize the risk of spillage. Primary surgery carries the risks of tumor thrombus with embolus and cardiac decompensation. In 1970’s Wagget and Koop firstly suggested the use of neoadjuvant chemotherapy and radiotherapy in WT for providing tumor thrombus regression (19).
After this report, some patients received neoadjuvant chemotherapy followed by surgery whereas some patients underwent primary nephrectomy. The patients with thrombus located in the infrahepatic inferior vena cava can underwent primary resection while the patients located above the liver can usually require cardiopulmonary bypass (CPB).
Now, Both SIOP group and children’s oncology group/NTWSG recommend preoperative chemotherapy to a patient presenting with intravascular tumor extension. SIOP approach is helpful to decrease the extent of the vascular thrombus which facilitates surgical excision. Most intracaval and intraatrial thrombi in WT show a response to chemotherapy (Table 1). Neoadjuvant chemotherapy causes tumor regression in nearly half of the patients. Hadley et al. reported that there was a measurable tumor regression in 47% of their patients and in 30% of those with atrial thrombus (7). After preoperative chemotherapy, three children with atrial thrombus managed without use of CPB. Similarly, Cristofani et al. reported that preoperative chemotherapy was able to reduce thrombus extension 8/11 (73%) treated patients. Surgical resection of tumor and thrombus was successful in their cases, receiving or not preoperative chemotherapy. They resolve completely after chemotherapy or can be removed through a cavotomy (18).
Use of preoperative chemotherapy reduces the size of the tumor and provides easy surgical removal. WT with tumor thrombus can be managed without the need for cardiac bypass by preoperative chemotherapy. Even complete dissolution of tumor thrombus is possible. Very few cases require exploration under CPB.
Aspiazu et al. shared their experience and outcomes of children with WT and tumor thrombus. In their group, six of seven patients presented with tumor thrombus extending into the right atrium and only one patient with infrahepatic thrombus. After receiving neoadjuvant chemotherapy, tumor thrombus regression was observed in all cases. Nephrectomy and thrombectomy were performed in all cases, requiring CPB for the four patients with suprahepatic and right atrial thrombus (6).
As a result of preoperative chemotherapy, the size of tumor thrombus can decrease and thrombectomy and nephrectomy becomes easier and safer. But, if the thrombus invades the vessel wall, cavectomy is recommended as a treatment option. Riberiora et al. reported their experience in three patients treated with cavectomy and they concluded that cavectomy is a safe procedure for treating pediatric patients with WT (20). If trombectomy is not possible, cavectomy can be preffered as a good surgical strategy. Generally, it has been recommended that if there is a renal tumor with intracaval tumor extension but without atrial extension, preoperative chemotherapy should be tried to shrinkage the tumor and tumor thrombus, firstly. After eight weeks of chemotherapy if the tumor still remains in the atria, it should be resected instead of persisting more chemotherapy.
Extension of the tumor to the retrohepatic vena cava or above would have required CPB and circulatory arrest to allow complete excision. CPB facilitates thrombus extraction in patients with suprahepatic vena caval and atrial extension. CPB is a complicated surgical procedure including the use of median sternotomy, atriatomy and systemic anticoagulation. It causes a longer operative time, higher transfusion amount and longer hospital stay.
Particularly, if atrial extension is still present after preoperative chemotherapy, most patients die with disease progression (7,10). Akyüz et al. reported that 14 patients received preoperative chemotherapy consisting of two-drug regimen. Akyüz et al. observed a high number of recurrences in children who had residual tumor thrombus. They concluded that surgical removal of the thrombus should be considered in the presence of life-threatening tumor thrombosis at diagnosis and in patients who had residual thrombus after chemotherapy (10). This finding shows the importance of complete resection. It seems that complete resection of all macroscopic disease should be tried. It has been recommended an aggressive approach to the management of patients with chemoresistant vena caval extension of their tumor. Murthi et al. reviewed their experience with surgical management of WT with persistent tumor thrombus. They used different approaches for excision of intravascular component such as local cavotomy in 3, extensive infra hepatic cavotomy without CPB in one and excision of cavoatrial tumor with CPB in nine patients (17).
Szavay et al. operated 24 patients who had tumor thrombus into the vena cava, in nine patients the thrombus reached into the right atrium. They used CPB in nine patients (15).
Presence of tumor thrombus does not cause a worse outcome if treated properly with preoperative chemotherapy and delayed resection. However, the presence of tumor thrombus increases the complication of primary resection. Complications are more frequent in the children with atrial extension than in those with extension limited to the inferior vena cava. The prognosis in children with WT and tumor thrombus is not affected by the presence or location of intravascular tumor thrombus, but an increased frequency of surgical complications has been reported. Chiappini et al. reported their results regarding 13 patients with an infradiaphragmatic tumor with thrombosis of the vena cava, the right atrium, or both underwent surgical resection with CPB, arrested circulation, and profound hypothermia. Postoperative complications were found in two patients (21).
In NTWSG-4, complications related to nephrectomy and tumor thrombectomy has been reported as 17.2% of cases with inferior vena cava extension and 36.7% of cases with atrial extension of tumor thrombus. Preoperative chemotherapy can lower the complication rate to 13%. The SIOP group reports 18% of complication in children with WT and thrombus extension (14,15). In children with extensive vascular invasion, particularly into the atrium, the best approach may be primary chemotherapy and delayed resection to minimize the risk of surgical complications.
It has been known that SIOP group recommends preoperative chemotherapy for most patients with presumed diagnosis of WT. Similarly, COG group also recommend preoperative chemotherapy for only patients with tumor thrombus. Preoperative chemotherapy has some advantages when compared to the initial surgery group such as shorter operative times, blood loss, and days of hospitalization.
But, the use of preoperative chemotherapy for patients with tumor thrombus has also some risks such as pulmonary embolism or tumor progression during preoperative chemotherapy. Vaideeswar et al. reported a case of sudden death due to hemorrhagic expansion of the intra-atrial component of WT after initiating of preoperative chemotherapy (22).
Schamberger et al. reported that complications during preoperative chemotherapy were seen in 5 of 69 patients such as tumor embolism, tumor progression (8).
Therefore, the decision of initial surgery or preoperative chemotherapy should be carefully determined on every case. Primary surgery would only be indicated in a patient who is unstable because of thrombus that might dislodge and cause acute symptoms.
The large series from the SIOP and NTWS groups demonstrated that the patients with tumor thrombus have a similar relapse-free and overall survival rates compared to the patients without tumor thrombus.
In conclusion, presence of tumor thrombus needs for multidisciplinary care including pediatric oncologists, pediatric surgeons, and pediatric cardiac surgeons. Surgical excision of extensive tumor thrombosis is suggested in the case of life-threatening thrombosis even with CPB. Preoperative chemotherapy should be suggested in cases lacking clinical symptoms of thrombosis.
Acknowledgements
None.
Footnote
Conflicts of Interest: The author has no conflicts of interest to declare.
References
- Dome JS, Fernandez CV, Mullen EA, et al. Children’s Oncology Group’s 2013 Blueprint for research: renal tumors. Pediatr Blood Cancer 2013;60:994-1000. [PubMed]
- Chintagumpala M, Muscal JA. Presentation, diagnosis, and staging of Wilms tumor. Up To Date, Ed, Pappo AS, 2013.
- Khozeimeh N, Sinha P, Dome JS, et al. Strategy for management of retroperitoneal tumors with caval tumor thrombus. J Pediatr Surg 2011;46:2065-70. [PubMed]
- Bader MI, Abdelaal K, Rogers T, et al. A surgical approach to Wilms’ tumour with retrohepatic vena caval extension. Pediatr Surg Int 2013;29:229-32. [PubMed]
- Xu B, Zhang Q, Jin J. Wilms tumor with renal vein tumor thrombus treated with only 3-port retroperitoneal laparoscopic technique. Urology 2013;81:1346-8. [PubMed]
- Aspiazu D, Fernandez-Pineda I, Cabello R, et al. Surgical management of Wilms tumor with intravascular extension: a single-institution experience. Pediatr Hematol Oncol 2012;29:50-4. [PubMed]
- Hadley GP, Sheik-Gafoor MH, Buckels NJ. The management of nephroblastoma with cavo-atrial disease at presentation: experience from a developing country. Pediatr Surg Int 2010;26:1169-72. [PubMed]
- Shamberger RC, Ritchey ML, Haase GM, et al. Intravascular extension of Wilms tumor. Ann Surg 2001;234:116-21. [PubMed]
- Lall A, Pritchard-Jones K, Walker J, et al. Wilms tumor with intracaval thrombus in the UK Children’s Cancer Study Group UKW3 trial. J Pediatr Surg 2006;41:382-7. [PubMed]
- Akyüz C, Emir S, Büyükpamukçu N, et al. Cavoatrial tumor extension in children with Wilms tumor: a retrospective review of 17 children in a single center. J Pediatr Hematol Oncol 2005;27:267-9. [PubMed]
- Abdullah Y, Karpelowsky J, Davidson A, et al. Management of nine cases of Wilms tumour with intracardiac extension-a single centre experience. J Pediatr Surg 2013;48:394-9. [PubMed]
- Cristofani LM, Duarte RJ, Almeida MT, et al. Intracaval and intracardiac extension of Wilms tumor. The influence of preoperative chemotherapy on surgical morbidity. Int Braz J Urol 2007;33:683-9. [PubMed]
- Murthi GV, Kocyildirim E, Sellathury S, et al. Wilms tumor with persistent intravascular extension: a review of the surgical aspects of management. J Pediatr Urol 2006;2:439-45. [PubMed]
- Ritchey ML, Kelalis PP, Haase GM, et al. Preoperative therapy for intracaval and atrial extension of Wilms tumor. Cancer 1993;71:4104-10. [PubMed]
- McDonald K, Duffy P, Chowdhury T, et al. Added value of abdominal cross-sectional imaging (CT or MRI) in staging of Wilms tumors. Clin Radiol 2013;68:16-20. [PubMed]
- Khanna G, Rosen N, Anderson JR, et al. Evaluation of diagnostic performance of CT for detection of Tumor thrombus in children with Wilms tumor: a report from the Children’s Oncology Group. Pediatr Blood Cancer 2012;58:551-5. [PubMed]
- Szavay P, Luithle T, Semler O, et al. Surgery of cavoatrial tumor thrombus in nephroblastoma: a report of the SIOP/GPOH. Pediatr Blood Cancer 2004;43:40-5. [PubMed]
- Refaie HD, Sarhan M, Hafez A. Role of CT in assesment of unresectable Wilms’ tumor response after preoperative chemotherapy in pediatrics. ScientificWorldJournal 2008;8:661-9. [PubMed]
- Wagget J, Koop CE. Wilms tumor: preoperative radiotherapy and chemotherapy in the management of massive tumors. Cancer 1970;26:338-40. [PubMed]
- Ribeiro RC, Schettini ST, Abib Sde C, et al. Cavectomy for the treatment of Wilms tumor with vascular extension. J Urol 2006;176:279-83; discussion 283-4. [PubMed]
- Chiappini B, Savini C, Marinelli G, et al. Cavoatrial tumor thrombus: single stage surgical approach with profound hypothermia and circulatory arrest, including a review of the literature. J Thorac Cardiovasc Surg 2002;124:684-8. [PubMed]
- Vaideeswar P, Chaudhari JP. Wilms tumor with right heart extension: report of a post-chemotherapeutic fatality. Indian J Pathol Microbiol 2012;55:381-3. [PubMed]


