
Intra- and juxta-articular osteoid osteoma in children and adolescents
Introduction
Osteoid osteoma (OO) is a relatively common benign tumor in children and adolescents, and the lesion is about two times more common in boys (1,2). Most OO occur preferentially in the metaphyseal or diaphyseal region of the long bone but have also been reported in almost every bone. Intra- and juxta-articular lesions are less common and have primarily only been included in case reports (3,4), with the most common manifestations being in the hip joint (5). In contrast, there are numerous studies on diaphysis OO with large case numbers (6,7). The clinical manifestations are most reported having the night pain alleviated with non-steroidal anti-inflammatory drugs (NASIDs) (8). Conventional radiographs, computed tomography (CT), magnetic resonance imaging (MRI), and technetium-99m bone scans are also necessary to correct diagnosis. Once diagnosed, the techniques reported having high success rates are percutaneous CT-guided radiofrequency ablation, CT-guided percutaneous excision, open or arthroscopic excision, and laser ablation (9-14).
Missing or delayed diagnosis may arise when atypical pain in connection with unusual radiological imaging modalities. Especially, juxta- and intra-articular OO may resemble more common entities such as the joint's traumatic or soft tissue pathologies. As a consequence, delayed diagnosis is the main cause of muscle atrophy, tenderness, local swelling or contracture (15). In fact, depended on the localization, especially joint pain, which is non-responsive to conventional treatment, many initial presumptive diagnoses must be reconsidered. With the advance of radiological techniques, the detective abilities of OO are significantly improved. Problems may arise in connection with a small lesion in and outside the joint that are not obvious. Due to the above problems, there are few studies investigating the presentation and treatment of OO about the difference between intra- and juxta-articular, which may frequently be mistaken for alternative articular pathologies.
In this study, we retrospectively reviewed intra- and juxta-articular OO in our hospital with a follow-up time of at least 2 years. The purpose of this study is to investigate the distinctions of the clinical presentation, imaging, diagnosis, and treatment between intra- and juxta-articular OO, with an emphasis on avoiding delaying of diagnosis and optimizing treatment strategies. We present the following article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-21-612/rc).
Methods
Patients
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by institutional ethics board of Children’s hospital of Fudan University [No. (2020) 128] and individual consent for this retrospective analysis was waived. A retrospective review was performed in all patients diagnosed with OO within or around joints from January 2008 to May 2019. Medical history was recorded, the inclusion criteria were: (I) a diagnosis of OO was confirmed involving the intra- and juxta-articular; (II) the follow-up time was longer than 24 months. The exclusion criteria were:
- the location of OO was on diaphysis;
- insufficient radiographic or clinical/pathology records;
- the follow-up time was less than 12 months.
Demographic and clinical data were collected. The time between complaints and diagnosis, the physical examination, the outcomes and complications, and the radiological finding were recorded.
Treatment procedure
The protocol of patients with pain of limbs in our center was as following:
- Physical examination was performed in connection with complaints.
- An X-ray was taken to evaluate if the obvious nidus existed (including cortical thickening, sclerosis, and a radiolucent nidus).
- CT/MRI and bone scan were needed when the radiograph positive; otherwise, resting and regular review.
- Surgeries were performed.
The OO was confirmed pathologically. The conception of juxta-articular was around capsular within metaphysis. The surgeries were all open excision. The interval time was the length from the first visit to diagnosis.
Statistical analysis
An Independent sample Student t-test was used to compare the continuous variables that fit the normal distribution between the two groups. The Mann-Whitney U test is used for variables that are not normally distributed. Chi-square analysis and Fisher’s exact test were used to compare categorical and bivariate variables. A P value <0.05 was considered significant for all statistical tests. Statistical analysis was performed using SPSS 19.0 (IBM, America).
Results
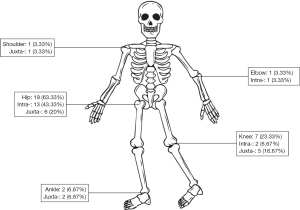
Thirty children and adolescents, 27 (90%) males and 3 (10%) females, were included and confirmed by pathological histology. The mean age at diagnosis was 8.37±3.79 years old, ranging from 2.3 to 15. The mean interval time was 4.67±5.88 months, ranging from 0 to 24 months. The mean follow-up time was 3.57±2.18 years (range, 1–10 years) (Table 1). The location of OO was shown in Figure 1. The most prominent complaints were pain, including night pain (60%), pain with activities (46.67%). Limping (76.67%) and abnormal range of motion of joint (53.33%) was also conspicuous.
Table 1
| Characteristic | Data |
|---|---|
| Sex (male/female) | 9:1 |
| Age at diagnosis (years) | 8.37±3.79 |
| Interval time (months) | 4.67±5.88 |
| Follow-up time (years) | 3.57±2.18 |
| X-ray (n) | 30 (100%) |
| CT (n) | |
| 1 time | 26 (86.67%) |
| 2 times | 3 (10%) |
| 3 times | 1 (3.33%) |
| MRI (n) | |
| 0 | 6 (20%) |
| 1 time | 22 (73.33%) |
| 2 times | 2 (6.67%) |
| Bone scan (n) | 27 (90%) |
| Complications (n) | |
| Secondary surgeries | 6 (20%) |
| Coxa vara | 1 (3.33%) |
CT, computerized tomography; MRI, magnetic resonance imaging.
On radiological data, all patients had X-ray, CT, or MRI. The cortical thickening and typical nidus were visible only in 12 (40%) patients on X-ray, 26 (86.67%) patients with a diagnostic lesion on CT for one time, 3 (10%) patients for two times, and 1 (3.33%) patient for three times to identify the disease. MRI was performed to observe the lesion, retinacular thickening, synovitis, and effusion. 2 (6.67%) patients even had MRI two times to find the lesion. In the 27 (90%) bone scans available for review, 24 (88.89%) were having an increased radiotracer uptake.
The initial diagnosis was based on the radiological data and clinical manifestation in 15 (50%) patients. However, no obvious factors were related to the alternative or delayed diagnosis. Factors identified on analysis to be significantly associated with intra- and juxta-articular OO, including pain with activity (P=0.004) and abnormal range of motion (P=0.00). Noticeably, the factor of abnormal range of motion (P=0.03) also influenced the existence of complications, including multiple surgeries, relapse, coxa vara, and limited range of motion (Table 2).
Table 2
| Variables | Total | Intra-articular | Juxta-articular | P | Complications | No complications | P | Delayed diagnosis | No delay | P |
|---|---|---|---|---|---|---|---|---|---|---|
| Patients (n) | 30 | 17 | 13 | – | 7 | 23 | 15 | 15 | – | |
| Sex (n) | 30 | 0.56 | 1 | 1 | ||||||
| Male | 27 | 16 | 11 | 6 | 21 | 14 | 13 | |||
| Female | 3 | 1 | 2 | 1 | 2 | 1 | 2 | |||
| Age (years) | 8.37±3.8 | 8.29±3.95 | 8.47±3.73 | 0.9 | 10.21±4.18 | 7.8±3.57 | 0.144 | 7.57±3.74 | 9.17±3.8 | 0.255 |
| Delayed time (months) | 4.67±5.88 | 3.76±4.16 | 3.08±4.96 | 0.9 | 5.43±4.69 | 2.87±4.31 | 0.47 | NA | NA | NA |
| Pain at night (n) | 30 | 1 | 1 | 0.06 | ||||||
| Yes | 18 | 10 | 8 | 4 | 14 | 12 | 6 | |||
| No | 12 | 7 | 5 | 3 | 9 | 3 | 9 | |||
| Pain with activity (n) | 30 | 0.004 | 0.66 | 0.27 | ||||||
| Yes | 14 | 12 | 2 | 4 | 10 | 9 | 5 | |||
| No | 16 | 5 | 11 | 3 | 13 | 6 | 10 | |||
| Abnormal range of motion (n) | 30 | 0 | 0.03 | 0.25 | ||||||
| Yes | 16 | 15 | 1 | 5 | 11 | 10 | 6 | |||
| No | 14 | 2 | 12 | 2 | 12 | 5 | 9 | |||
| NSAIDs (n) | 30 | 1 | 1 | 0.22 | ||||||
| Yes | 3 | 2 | 1 | 0 | 3 | 3 | 0 | |||
| No | 27 | 15 | 12 | 7 | 20 | 12 | 15 | |||
| Delayed diagnosis (n) | 30 | 0.14 | 0.39 | NA | ||||||
| Yes | 15 | 11 | 4 | 5 | 10 | |||||
| No | 15 | 6 | 9 | 2 | 13 | |||||
| Complications (n) | 30 | 0.1 | NA | 0.39 | ||||||
| Yes | 7 | 6 | 1 | 5 | 2 | |||||
| No | 23 | 11 | 12 | 10 | 13 | |||||
| Articular (n) | 30 | NA | 0.1 | 0.14 | ||||||
| Yes | 17 | 6 | 11 | 11 | 6 | |||||
| No | 13 | 1 | 12 | 4 | 9 | |||||
| Size of tumor (CT) (mm3) | 398.37± 378.94 |
456.71± 461.09 |
322.08± 228.95 |
0.385 | 445.57± 632.82 |
384± 281.32 |
0.44 | 289.47± 169.72 |
507.27± 493.18 |
0.16 |
NA, not available; NSAIDs, non-steroidal anti-inflammatory drugs; CT, computerized tomography.
Six children (20%) with intra-articular lesions, underwent surgical resection after a diagnosis of OO, but they developed limb pain again on average 1-year later, and further examination suggested recurrence of OO. Therefore, the second operation was warranted to remove the lesion again. Fortunately, no one needed the third operation.
Discussion
Jaffe introduced OO on 5 cases of benign bone tumor in 1935 (16). Approximately one out of ten cases of OO are intra- or juxta-articular (4). It is not difficult to recognize the regular lesion based on the characteristic clinical, radiological and histological pictures. However, it is still a challenge if the position is located in the joint capsules, which may produce a variety of nonspecific manifestations and confusing radiological features (5).
OO occurs predominantly in the appendicular skeleton. According to the Musculoskeletal Tumor Society staging system, the lesion was classified as cortical, cancellous, and subperiosteal (17,18). The hip was the most commonly affected joint, accounting for 28–40% of all cases (19). In our study, lesions were seen in most of the functional joints, with the hip being the most prominent (63.33%). The tricking point of intra- and juxta-articular lesions lies in their insidious clinical manifestations, which leads to delayed diagnosis and thus makes the children’s pain unable to be resolved. It was worth noticing that the physis lesions can cause limb length discrepancy with potential coronal and/or sagittal malalignment.
Pain is the most common symptom, even the lesion involved joints (20). The clinical manifestation can be subtle, such as the patient can present with referred pain in the knee if the lesion is involved in the hip. Synovitis is another feature when the articular lesion continues to progress. Joint pain, flexion contracture, decreased range of motion, and a limp or antalgic gait can also be seen in these patients (15). Problems in the confused diagnosis may arise in connection with an unusual location, since it causes atypical clinical manifestations (4). According to the previous clinical features, the common pitfalls are Osteomyelitis/intraosseous abscess, fracture/stress reaction, osteoblastoma and chondroblastoma (21,22). A history of injury can make the diagnosis of OO more difficult, especially if symptoms appear shortly after injury. In the present study, most patients presented with pain as the primary symptom, although the location was variable and influenced by status. However, loss of function took the form of decreased range of motion of the affected joint, including a limp, abnormal range of motion, and uncomfortable when playing. Pain with activity and abnormal range of motion was significantly different between intra- and juxta-articular patients. This may attribute to the constant friction on the intra-articular lesion. More severe synovitis can cause more pronounced pain; other than that, long-standing inflammation may damage the cartilage on the joint surface (6,15). From that, consistently abnormal range of motion also obviously influenced the complications. Therefore, we suggest that in the case of suspected intra-articular cases, the relevant examinations should be actively improved to avoid permanent damage to the intra-articular structures.
Plain radiography is warranted when children are complaining of pain or limited range of motion. The manifestation is different from the traditional cortical lesion, and the surrounding reactive sclerosis can be minimal or absent in intra- and juxta-articular OO (23). The nidus is not visualized on plain films, but additional findings such as loss of function often remind the physician to take further imaging workup. The sensitivity of Technetium-99-labeled bone scintigraphy is 100% for confirming the diagnosis of OO (24). So, if the bone scan imaging is positive, limited-field, thin-cut CT scans should be the next imaging modality for more precise localization. CT is the modality of choice for diagnosis and specifying the location of the lesion. In the present study, all patients had CT scans, but not all patients had the classical image of the nidus, so multiple CTs was performed. The reasons why we must do CT are as follows: (I) CT can help us locate the lesion accurately and facilitate subsequent surgical operations; (II) CT can exclude other diseases with similar clinical manifestations, such as bone tuberculosis, infection, etc. Given the exposure of ionizing radiation, the MRI might be preferred to CT, especially in the pediatric population (21). MRI is also a reliable method of visualizing the nidus since it is more sensitive to detecting reactive changes in soft tissue. It is also better to evaluations with a small field of view on the axial plane and proton density sequences (25). Germann et al. (26) claimed that MRI was excellently suited for diagnosing intra- and extra-articular OO because the joint effusion and synovitis were distinguished between the intra- and extra-articular. However, it was not easy to separate the soft tissue edema and synovitis from intra- and juxta-articular OO.
Although non-operative treatment can be considered as an option since some patients might heal spontaneously (27), in our center, all patients underwent surgical resection since symptoms persisted, even with NSAIDs. Moreover, in children with growth potential, the continued presence of these tumors can lead to undesired complications, such as limb length discrepancies or osteoarthritis. In this study, six patients (13.33%) had performed secondary surgeries (Figure 2). The reasons were as following: (I) the resection was incomplete; (II) The location of the lesion was inaccurate during the operation; More physicians proposed to perform excision for intra-articular locations of the tumor (23,28-31). Over the years, CT-guided techniques, including trephine excision, cryoablation, radiofrequency ablation, and laser thermocoagulation, were reported to be a successful and low risk (32-35). However, although this procedure is minimally invasive, it entails some problems, such as it is difficult to obtain an intraoperative tumor specimen, which is still a gold stand for definitive diagnosis of OO on histological examination (36). Recently, a literature review reported that arthroscopic management of OO of the upper extremity joints is highly successful and results in no tumor recurrence (23). As for the hip lesion, arthroscopic management appears to be an effective method (37), but has a high revision rate (10/25) (38). There is a risk of incomplete resection in areas more difficult to access by minimally invasive procedures, such as arthroscopy, and may also lead to neurovascular injuries in areas that are near important anatomic structures (39).

In growing children and adolescents, intra- and juxta-articular OO may cause skeletal abnormalities, including angular deformity of the long bone and leg length discrepancy (40). The lesion located in femoral neck may increase femoral antetorsion and neck-shaft angle (41), and may contribute to a Cam-type deformity (42). Most skeletal sequelae in children are expected to disappear or improve after treatment, however, skeletal hypertrophy around the joint may lead to permanent abnormalities, such as limited range of joint motion (15).
The present study had limitations. First, this is a retrospective study to investigate the distinctions between intra- and juxta-articular OO; more randomized controlled trials or prospective studies are required for further validation. Second, the number of patient samples in this study is too small, and a large sample study should be added for verification. Third, all patients need longer follow-up time until skeletal maturity at the final follow-up to confirm if there are any recurrences and limb deformities.
In conclusion, Intra- and juxta-articular OO has atypical clinical and radiographic features, clinicians should keep in mind the possibility of this disorder and conduct a further examination, as the delayed diagnosis can lead to unnecessary pain and psychological disorders in children. It is crucial to identify the location of OO when children have pain with activity and abnormal range of motion. The persistently abnormal range of motion also significantly leads to complications. The open surgeries to resect the nidus in juxta-articular OO were effective.
Acknowledgments
Funding: The project was sponsored by Shanghai Shenkang Clinical Research Cultivation Project (2019) (No. SHDC12019X28), Natural Science Foundation of Shanghai (No. 20Y11912900) and the Clinical Center of Birth Defects (No. EK2022ZX03) of Children’s Hospital of Fudan University.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-21-612/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-21-612/dss
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-21-612/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by institutional ethics board of Children’s Hospital of Fudan University [No. (2020) 128] and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Healey JH, Ghelman B. Osteoid osteoma and osteoblastoma. Current concepts and recent advances. Clin Orthop Relat Res 1986;76-85. [Crossref] [PubMed]
- Peyser A, Applbaum Y, Simanovsky N, et al. CT-guided radiofrequency ablation of pediatric osteoid osteoma utilizing a water-cooled tip. Ann Surg Oncol 2009;16:2856-61. [Crossref] [PubMed]
- Gaeta M, Minutoli F, Pandolfo I, et al. Magnetic resonance imaging findings of osteoid osteoma of the proximal femur. Eur Radiol 2004;14:1582-9. [Crossref] [PubMed]
- Franceschi F, Marinozzi A, Papalia R, et al. Intra- and juxta-articular osteoid osteoma: a diagnostic challenge: misdiagnosis and successful treatment: a report of four cases. Arch Orthop Trauma Surg 2006;126:660-7. [Crossref] [PubMed]
- May CJ, Bixby SD, Anderson ME, et al. Osteoid Osteoma About the Hip in Children and Adolescents. J Bone Joint Surg Am 2019;101:486-93. [Crossref] [PubMed]
- Boscainos PJ, Cousins GR, Kulshreshtha R, et al. Osteoid osteoma. Orthopedics 2013;36:792-800. [Crossref] [PubMed]
- Iyer RS, Chapman T, Chew FS. Pediatric bone imaging: diagnostic imaging of osteoid osteoma. AJR Am J Roentgenol 2012;198:1039-52. [Crossref] [PubMed]
- Bottner F, Roedl R, Wortler K, et al. Cyclooxygenase-2 inhibitor for pain management in osteoid osteoma. Clin Orthop Relat Res 2001;258-63. [Crossref] [PubMed]
- Hoffmann RT, Jakobs TF, Kubisch CH, et al. Radiofrequency ablation in the treatment of osteoid osteoma-5-year experience. Eur J Radiol 2010;73:374-9. [Crossref] [PubMed]
- Lindner NJ, Ozaki T, Roedl R, et al. Percutaneous radiofrequency ablation in osteoid osteoma. J Bone Joint Surg Br 2001;83:391-6. [Crossref] [PubMed]
- Mylona S, Patsoura S, Galani P, et al. Osteoid osteomas in common and in technically challenging locations treated with computed tomography-guided percutaneous radiofrequency ablation. Skeletal Radiol 2010;39:443-9. [Crossref] [PubMed]
- Peyser A, Applbaum Y, Khoury A, et al. Osteoid osteoma: CT-guided radiofrequency ablation using a water-cooled probe. Ann Surg Oncol 2007;14:591-6. [Crossref] [PubMed]
- Fenichel I, Garniack A, Morag B, et al. Percutaneous CT-guided curettage of osteoid osteoma with histological confirmation: a retrospective study and review of the literature. Int Orthop 2006;30:139-42. [Crossref] [PubMed]
- Roqueplan F, Porcher R, Hamzé B, et al. Long-term results of percutaneous resection and interstitial laser ablation of osteoid osteomas. Eur Radiol 2010;20:209-17. [Crossref] [PubMed]
- Song MH, Yoo WJ, Cho TJ, et al. Clinical and radiological features and skeletal sequelae in childhood intra-/juxta-articular versus extra-articular osteoid osteoma. BMC Musculoskelet Disord 2015;16:3. [Crossref] [PubMed]
- Jaffe HL. Osteoid-osteoma: a benign osteoblastic tumor composed of osteoid and atypical bone. Arch Surg 1935;31:709-28. [Crossref]
- Kitsoulis P, Mantellos G, Vlychou M. Osteoid osteoma. Acta Orthop Belg 2006;72:119-25. [PubMed]
- Noordin S, Allana S, Hilal K, et al. Osteoid osteoma: Contemporary management. Orthop Rev (Pavia) 2018;10:7496. [Crossref] [PubMed]
- Campanacci M, Ruggieri P, Gasbarrini A, et al. Osteoid osteoma. Direct visual identification and intralesional excision of the nidus with minimal removal of bone. J Bone Joint Surg Br 1999;81:814-20. [Crossref] [PubMed]
- Bauer TW, Zehr RJ, Belhobek GH, et al. Juxta-articular osteoid osteoma. Am J Surg Pathol 1991;15:381-7. [Crossref] [PubMed]
- Carneiro BC, Da Cruz IAN, Ormond Filho AG, et al. Osteoid osteoma: the great mimicker. Insights Imaging 2021;12:32. [Crossref] [PubMed]
- Chai JW, Hong SH, Choi JY, et al. Radiologic diagnosis of osteoid osteoma: from simple to challenging findings. Radiographics 2010;30:737-49. [Crossref] [PubMed]
- Ge SM, Marwan Y, Abduljabbar FH, et al. Arthroscopic management of intra- and juxta-articular osteoid osteoma of the upper extremity: a systematic review of the literature. Eur J Orthop Surg Traumatol 2020;30:1333-44. [Crossref] [PubMed]
- Wells RG, Miller JH, Sty JR. Scintigraphic patterns in osteoid osteoma and spondylolysis. Clin Nucl Med 1987;12:39-44. [Crossref] [PubMed]
- Davies M, Cassar-Pullicino VN, Davies AM, et al. The diagnostic accuracy of MR imaging in osteoid osteoma. Skeletal Radiol 2002;31:559-69. [Crossref] [PubMed]
- Germann T, Weber MA, Lehner B, et al. Intraarticular Osteoid Osteoma: MRI Characteristics and Clinical Presentation Before and After Radiofrequency Ablation Compared to Extraarticular Osteoid Osteoma. Rofo 2020;192:1190-9. [Crossref] [PubMed]
- Moberg E. The natural course of osteoid osteoma. J Bone Joint Surg Am 1951;33 A:166-70.
- Ramaswamy AG, Kumaraswamy V, Patil N, et al. Arthroscopic Excision of Osteoid Osteoma of the Femoral Neck. Indian J Orthop 2018;52:568-71. [Crossref] [PubMed]
- Kamrani RS, Moradi A, Sharafat Vaziri A, et al. Arthroscopic ablation of an osteoid osteoma of the elbow: a case series with a minimum of 18 months' follow-up. J Shoulder Elbow Surg 2017;26:e122-7. [Crossref] [PubMed]
- Ge SM, Marwan Y, Addar A, et al. Arthroscopic Management of Osteoid Osteoma of the Ankle Joint: A Systematic Review of the Literature. J Foot Ankle Surg 2019;58:550-4. [Crossref] [PubMed]
- Stražar K, Slodnjak I, Zupanc O, et al. Arthroscopic removal of osteoid osteoma with gamma probe assistance in the hip joint. Hip Int 2019;29:328-35. [Crossref] [PubMed]
- Esteban Cuesta H, Martel Villagran J, Bueno Horcajadas A, et al. Percutaneous radiofrequency ablation in osteoid osteoma: Tips and tricks in special scenarios. Eur J Radiol 2018;102:169-75. [Crossref] [PubMed]
- Tomasian A, Cazzato RL, Auloge P, et al. Osteoid osteoma in older adults: clinical success rate of percutaneous image-guided thermal ablation. Clin Radiol 2020;75:713.e11-6. [Crossref] [PubMed]
- Santiago E, Pauly V, Brun G, et al. Percutaneous cryoablation for the treatment of osteoid osteoma in the adult population. Eur Radiol 2018;28:2336-44. [Crossref] [PubMed]
- Bianchi G, Zugaro L, Palumbo P, et al. Interventional Radiology's Osteoid Osteoma Management: Percutaneous Thermal Ablation. J Clin Med 2022;11:723. [Crossref] [PubMed]
- Daniilidis K, Martinelli N, Gosheger G, et al. Percutaneous CT-guided radio-frequency ablation of osteoid osteoma of the foot and ankle. Arch Orthop Trauma Surg 2012;132:1707-10. [Crossref] [PubMed]
- Eberhardt O, von Kalle T, Matthis R, et al. A CT-free protocol to treat osteoid osteoma of the hip region in childhood and adolescence by percutaneous drilling and by hip arthroscopy. Hip Int 2021; [Epub ahead of print]. [Crossref] [PubMed]
- Dai L, Zhang X, Mei Y, et al. Arthroscopic Excision of Intra-Articular Osteoid Osteoma of the Hip: A Case Series. Arthroscopy 2021;37:3104-12. [Crossref] [PubMed]
- Nourissat G, Kakuda C, Dumontier C. Arthroscopic excision of osteoid osteoma of the elbow. Arthroscopy 2007;23:799.e1-4. [Crossref] [PubMed]
- Norman A, Dorfman HD. Osteoid-osteoma inducing pronounced overgrowth and deformity of bone. Clin Orthop Relat Res 1975;233-8. [Crossref] [PubMed]
- Giustra PE, Freiberger RH. Severe growth disturbance with osteoid osteoma. A report of two cases involving the femoral neck. Radiology 1970;96:285-8. [Crossref] [PubMed]
- Pianta M, Crowther S, McNally D, et al. Proximal femoral intra-capsular osteoid osteoma in a 16-year-old male with epiphyseal periostitis contributing to Cam-type deformity relating to femoro-acetabular impingement. Skeletal Radiol 2013;42:129-33. [Crossref] [PubMed]


