No more pediatric code blues on the floor: evolution of pediatric rapid response teams and situational awareness plans
Reducing or eliminating cardiac or respiratory arrests (code blues) that occur on the inpatient, noncritical care units of children’s hospitals is an undoubtedly desirable goal. In an ideal world, pediatric medical providers would prevent the occurrence of all pediatric cardiac arrests, but we know that this is not feasible as they occur in both low and high resource environments worldwide. Reducing these code blue events is not an easy task. A 20-hospital collaborative tried to substantially reduce their code events outside of their intensive care units (ICUs) by implementing a package of process improvement strategies. The collaborative was only able to show partial improvement in a subset of the hospitals a year after the conclusion of data collection (1). This presents a challenge for ongoing efforts to move pediatric codes from noncritical care areas to ICUs, and as demonstrated, will likely take time, coordination and standardization to achieve. We will focus on two different global targets of improvement: the first is the elimination or reduction of “preventable” codes, and the second is assuring the highest quality resuscitation response for those children that do suffer an unavoidable arrest. As we will discuss, this should optimally occur in the pediatric intensive care unit (PICU).
Pediatric in-hospital cardiac arrest is an uncommon and unwanted sequela of critical illness. Though the outcomes of pediatric cardiac arrest have generally improved over the past thirty years, the incidence and outcomes continue to prove variable in the literature. Recent studies have demonstrated an incidence of cardiac arrest in hospitalized pediatric patients of 0.7–3% (2,3) with survival to discharge rates ranging from 13.7–34% (2-5).
Though outcomes for in-hospital cardiac arrests have long been established as superior to outcomes for out-of-hospital cardiac arrests (6,7), information is limited regarding the effect of hospital location on outcomes after such pediatric cardiac arrests. As the incidence of cardiac arrest is significantly lower on the wards when compared to the PICU (3,5,8), intuitively, one would assume that the effectiveness of resuscitation in critical care areas would be superior to that on the wards. This can reasonably be hypothesized to be related to the availability of increased monitoring, increased resources, more highly trained personnel, and greater familiarity of the team with the resuscitation process given the higher frequency of events in critical care areas.
In one retrospective single-center study of in-hospital cardiac arrests over a five-year period at a tertiary care pediatric hospital in Finland, the location of cardiac arrest did not affect outcome (2). A prospective single-center study of in-hospital cardiac arrests at a pediatric tertiary care hospital in Australia similarly demonstrated no difference in outcomes for patients on wards versus the ICU at the time of the arrest (5). A multicenter study utilizing patient data from the Pediatric Emergency Care Applied Research Network (PECARN) demonstrated no difference in mortality based on in-hospital location at time of arrest (8). Alternatively, data from a tertiary care adult hospital in Italy suggested that location of in-hospital arrest affects survival to discharge; patients with cardiac arrests in “monitored” areas, defined as the emergency room and ICU, where a dedicated cardiac arrest team was immediately available, were significantly more likely to survive than those in “non-monitored” areas, defined as the wards and X-ray where the cardiac arrest team was not immediately available. Furthermore, arrests occurring in monitored areas had faster times to advanced life support, which independently correlated with improved survival to discharge (9).
Given the albeit under-validated concern that arrest resuscitations outside of the critical care areas are inferior, several studies have investigated strategies to improve the timeliness and quality of resuscitation efforts in out-of-ICU settings. An adult hospital in Finland was able to improve outcomes following cardiac arrests outside of critical care areas with the introduction of an ICU-based resuscitation team (10). Another group at a large, tertiary care pediatric hospital attempted implementation of formalized code team training, consisting of high-fidelity in-situ simulation sessions attended by all members of a multidisciplinary code team with a goal to improve American Heart Association guideline compliance in out-of-ICU cardiac arrests (11). Though there was subjective improvement in code team positioning and communication as well as some improvement in ventilation and chest compression rates following the training, the team was unable to achieve significant improvement in guideline adherence.
Despite the conflicting evidence to support improved outcomes of pediatric cardiac arrest in critical care areas versus the wards, the trend over time has been for pediatric code events to move towards the ICU. Data from the Get With the Guidelines® Resuscitation Registry showed from 2004 to 2011 the percentage of all in hospital pediatric cardiac arrests occurring in the ICU increased from 87–91% to 94–96% (12).
Moving the patient before they code: implementation of Pediatric Rapid Response Teams
In 1999, To Err is Human: Building a Safer Health System was published and revealed a staggering estimate of hospital patient deaths per year secondary to medical errors (13). These errors were reported to be the result of flawed system-based operations, and the authors implored hospitals to implement drastic, system-wide changes to prevent further morbidity and mortality. In response, the Institute for Healthcare Improvement launched the “100,000 Lives Campaign” in 2004 with a goal to save 100,000 hospitalized patient lives over 18 months through improvements in safety and effectiveness of healthcare. The primary intervention was to deploy rapid response teams, intended to identify hospitalized patients exhibiting signs of deterioration and allow for intervention before cardiopulmonary arrest occurs. The implementation of these teams was reported in the adult literature to not only improve mortality post cardiac arrest but also to reduce the incidence of unexpected cardiac arrests (9,14,15). Over the subsequent decade, pediatric rapid response teams were modeled after adult teams in an effort to similarly reduce the incidence and improve the outcomes of pediatric cardiac arrests on the wards.
Multiple studies have investigated the implementation of pediatric rapid response teams and their effect on pediatric cardiac arrest incidence and outcomes. Though many of the initial studies were criticized for demonstrating a reduction in ward cardiac arrests only, theorizing that patients were moved from the wards to the ICU only to suffer a cardiac arrest in a different location, several studies have demonstrated successful reduction in overall patient mortality. Implementation of a medical emergency team at a pediatric hospital in Melbourne, Australia in 2002 resulted in a trend toward reduction in both cardiac arrests and mortality over a one year period though neither reached statistical significance (16). The implementation of a Medical Emergency Team at Cincinnati Children’s Hospital Medical Center in 2005 resulted in a 59% decrease in all codes and a 73% decrease in “preventable” codes (17). The implementation of a rapid response team at Lucile Packard Children’s Hospital in 2005 demonstrated an 18% reduction in the mean monthly mortality rate hospital-wide as well as a reduction in mean monthly code rates outside of the pediatric ICU (18). Similarly, implementation of a rapid response system at a 136-bed pediatric hospital in the US in 2005 resulted in a reduction in cardiac arrests and duration of clinical instability before evaluation by critical care personnel (19). Alternatively, a prospective, multicenter trial examining deployment of a standardized pediatric rapid response team at four pediatric academic hospitals in Canada demonstrated a reduction in PICU mortality only after readmission, but no reduction in the rate of cardiopulmonary arrests or PICU mortality (20). Using the PHIS database, an interrupted time series analysis examined the effect of implementation of a rapid response team on annual risk-adjusted mortality rates at 38 freestanding pediatric hospitals and found no association with a reduction in hospital mortality (21).
Furthermore, the implementation of pediatric rapid response teams across institutions is not standardized, making comparisons challenging. A 2013 investigation of academic American pediatric hospitals demonstrated that while formal rapid response teams have become customary at such institutions, there was marked heterogeneity in activation triggers, team structure, response time and follow up (22). To date, there are no studies which demonstrate an optimal team composition and structure. However, data from one tertiary pediatric hospital in 2017 demonstrated that pediatric patients reviewed by rapid response teams were at significantly higher risk of mortality, and that this risk further increased for children with multiple rapid response team activations (23). Given that greater than 50% of second rapid response team activations occurred within 24 hours, the investigators suggested follow up for 24–48 hours given this identified and substantial risk.
In 2017, investigators used The Get With the Guidelines® Resuscitation Registry to examine the utilization of pediatric rapid response teams for patients who require cardiopulmonary resuscitation (24). They demonstrated that of 215 children from 23 hospitals who required CPR on the wards or within 48 hours of transfer to the ICU, only 22% had a preceding rapid response team evaluation; of those who did not have a preceding rapid response and for whom vital sign data was available, 39% had at least one abnormal vital sign that could have triggered an activation. This suggests that current rapid response teams could be better utilized to recognize patients with impending deterioration. While identification of a declining patient has long been touted as a critically honed skill of nurses, physicians, respiratory therapists and even family members, this data suggests that more than just individual assessments are needed to appropriately recognize the at-risk patient. It can further be concluded from this evidence that who and what triggers the activation of a rapid response team is a key component of successful rapid response process utilization.
Detecting potential deterioration: early warning or risk scores
The practice of when to call a rapid response team varies from institution to institution. Traditional vital sign triggers for physician notification weren’t classically intended to escalate to a rapid response team, nor was it the intent of the Institute of Medicine when they recommended the implementation of rapid response teams. Efforts to predict which patients are at risk of deterioration have generally taken the form of assigning scores to vital signs, laboratory markers and physical exam findings, and then combining those into a single score that yields some prediction of the risk of deterioration, now termed “early warning scores.” Because the rates of codes and deterioration in the pediatric population are so much smaller than the adult population, the yields of these scores tend not to be quite as fruitful as in the adult world, yet several scores have shown predictive values across a wide spectrum of pediatric illness.
The Pediatric Early Warning Score (PEWS) is the most commonly used and studied of these early warning scores though multiple variations of the scoring system exist. PEWS scoring successfully identified children at risk for cardiopulmonary arrest in an international, multicenter, case-control study in 2011 (25). Subsequent studies have shown that PEWS scoring is a better predictor than physician judgement of the risk of needing ICU transfer (26) and that PEWS can be applied across subspecialty patients (27,28). However, in 2017, a systematic review examining the evidence for PEWS use in the pediatric healthcare setting found heterogeneous implementation and limited standardization of outcomes making aggregate analysis impossible (29). The study pointed towards positive trends in clinical outcomes as a result of PEWS implementation, but the authors urged for utilization of PEWS within the context of larger safety system and for a more uniform approach to allow for true analysis of its impact.
A recently published international multicenter study randomized 21 hospitals providing care for pediatric patients across seven countries to implement PEWS or continue usual care without severity of illness scoring and subsequently compared all-cause hospital mortality (30). Though the study failed to show a decrease in codes or mortality, it is important to note that all hospitals underwent intensive training around situational awareness (SA), and all of the hospitals showed dramatic improvement in all-cause hospital mortality over the predicted numbers used in power calculations. One persistent question is whether mortality is the best endpoint for such studies as there is certainly morbidity associated with missing or delayed recognition of deterioration of pediatric patients. This study also raises the question of whether the focus should be less on objective scoring systems which may isolate patients to a “number” and instead encompass a larger culture of safety initiatives which include subjective assessments.
There are additional predictive scores being used or developed to help predict clinical deterioration. The Rothman Index (31) was developed to be a continuous update on the current status of a patient and has recently demonstrated validity in hospitalized pediatric patients. A modified version was tested against an in-house system of scoring based on vital sign classification at the Children’s Hospital of Pittsburgh (32), but did not show added value over the vital sign scoring. There are also institutional variations to the Pediatric Early Warning system that have been published in numerous studies to adapt to different settings or purposes.
Intangibles and SA: watchers, gut feelings, family concerns and other ways to tap into experience
The majority of pediatric inpatients recover from their illnesses and survive to discharge, never veering into the worrisome path of clinical deterioration. The challenge in pediatric inpatient medicine is to consistently identify the small subset of patients who are at risk. Moving even further, the goal should be to capture those patients proactively rather than reactively, ideally preventing deterioration rather than reversing it. Scoring systems do not capture 100% of patients who deteriorate, and so the question becomes how can we supplement scoring systems without overly burdening or making paranoid the providers and family?
SA is a term that comes from highly reliable organizations such as the nuclear power and airline industries. The concept of SA is an understanding of “knowing what’s going on,” and is defined as “the perception of elements in the environment within a volume of time and space, the comprehension of their meaning, and the projection of their status in the near future.” (33-35). In the healthcare setting this means not just having the data on the patient at hand, but rather synthesizing that data into an accurate picture of the patient’s clinical status and trajectory. It also then would involve acting upon that data when the trajectory is not following the desired path or deviates from what is expected. How to improve SA depends on the institution and the resources available there.
Some institutions, including the Ohio Children’s Hospitals’ Solutions for Patient Safety group, have examined the recently developing concept of “watchers”. The watcher is a patient that despite perhaps reassuring risk scores and/or vitals, makes some member of the team worried that they are going to deteriorate. This may be based on a “gut feeling” gained from experiences with similar patients in the past, an overall negative trajectory of the patient’s course, worrisome clinical traits not captured in a scoring system, or something about the constellation of findings that simply triggers a warning to providers. Perhaps the best source of which patients warrant “watching” is parental concern, particularly in the growing population of complex systems patients whose baselines are defined best by their home-based caregivers rather than by the traditional medical norms. It is for this, and other reasons, why some institutions include parental activation of the rapid response team as an option (36).
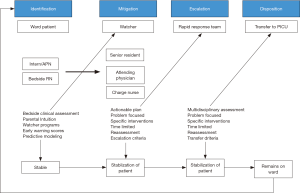
The incorporation of a watcher program into a clinical system has been, to date, institutionally-specific. While some institutions have a formalized watcher process with regularly scheduled assessments by team members for identified patients, other institutions identify watchers at daily SA huddles to increase cognizance of patients felt to be at risk. In one pediatric hospital, twice-daily multidisciplinary watcher huddles were introduced on each floor over a nine-month period (37). The huddles served to identify watcher patients on the basis of predefined criteria and develop mitigation plans for clinical changes. The subsequent 12 months demonstrated an 81% reduction in the proportion of UNSAFe (unrecognized situational awareness failure events) transfers to the PICU, defined as patients requiring intubation, pressor administration or greater than three fluid boluses within 1 hour of transfer. Figure 1 depicts the complex model for identification and escalation of care for at-risk patients.
Future
One of the limitations to the investigation and reduction of pediatric mortality is that the low rate of pediatric code events both in the ICU and on the floor, as well as the very low pediatric mortality rate, make end points in research challenging. To continue to make improvements in hospital systems, there need to be both process and outcome measures that have the potential to show change in a more concise timeline. Several groups have looked at “clinical deterioration events” as a surrogate, referring to patients who require near immediate escalation of care in the form of intubation, vasoactive medication administration, significant volume resuscitation or cardiopulmonary resuscitation within a designated time frame following transfer to the PICU (36,38). The idea behind this surrogate is to capture patients for whom prior recognition could have (I) predicted rapid deterioration and (II) allowed for successful resuscitation to prevent the decline. The classic example of the patient with smoldering sepsis who becomes febrile and tachycardic, who if not intervened upon promptly, progresses to hypotensive shock and cardiac arrest is an uncommon uncomplicated scenario in today’s medical arena of increasingly complex patients. Often, our patients’ stories are much harder to decipher, hence the challenges we face in identifying those whose stories lead to morbidity and mortality.
The hope for big data predictive modeling exists. With the advent of electronic medical records, the access to large quantities of data across vast numbers of admissions gives hope to tapping that data for predictive modeling. Recently an ensemble boosting model of predicting transfer to the ICU from such a database performed better than PEWS (39). Some caution should be taken with such predictive models given the marked heterogeneity in standards for PICU admission across institutions. One group of investigators at three large children’s hospitals developed a model using machine learning methods with many predictor variables to forecast those patients at greatest risk of a clinical deterioration event within 6 hours of transfer to the PICU (40). This clinical deterioration event is similar to the models described previously by Brady and Bonafide, but was specifically looking for patients meeting criteria for deterioration at specific time points after the evaluation point. The model was able to predict clinical deterioration 6 hours after evaluation at 80% sensitivity and 80.5% specificity, but with a positive predictive value of 5.2%. Though the numbers are not ideal, they indicate a potential for more accurate prediction and provide an opportunity for further study. Though a definitive benefit to parents and pediatric providers, the fact that kids are less likely to deteriorate than adults makes it harder to create a really high performing screening tool.
Summary
Minimizing code blues on the floor of children’s hospitals is an achievable goal. There are steps that every institution can take from the most fundamental level of interaction between a bedside nurse and a patient, all the way up to complex electronic systems that can help to identify and predict which patients are at the greatest risk for deterioration. Thereafter, institutions must intervene to decrease the risk for further deterioration and enable transfer to the PICU early enough so that if an arrest does happen it occurs within the ICU. These interventions start with improved monitoring and response to alarms, using a combination of both objective scoring systems and the subjective experience of seasoned staff to identify patients at risk. Once those patients are identified, the implementation of a robust system to address clinical concerns and ensure adequate resources are delivered to the patient becomes paramount. The inclusion of an escalation plan, which typically involves an efficient and effective rapid response team, minimizes barriers to transfer when necessary, and allows patients to get to the PICU in a more timely fashion. Finally, by optimizing the function of both rapid response and code teams, patients whose clinical course inevitably includes deterioration can have improved outcomes.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Hayes LW, Dobyns EL, DiGiovine B, et al. A multicenter collaborative approach to reducing pediatric codes outside the ICU. Pediatrics 2012;129:e785-91. [Crossref] [PubMed]
- Suominen P, Olkkola KT, Voipio V, et al. Utstein style reporting of in-hospital paediatric cardiopulmonary resuscitation. Resuscitation 2000;45:17-25. [Crossref] [PubMed]
- Reis AG, Nadkarni V, Perondi MB, et al. A prospective investigation into the epidemiology of in-hospital pediatric cardiopulmonary resuscitation using the international Utstein reporting style. Pediatrics 2002;109:200-9. [Crossref] [PubMed]
- Slonim AD, Patel KM, Ruttimann UE, et al. Cardiopulmonary resuscitation in pediatric intensive care units. Crit Care Med 1997;25:1951-5. [Crossref] [PubMed]
- Tibballs J, Kinney S. A prospective study of outcome of in-patient paediatric cardiopulmonary arrest. Resuscitation 2006;71:310-8. [Crossref] [PubMed]
- Moler FW, Meert K, Donaldson AE, et al. In-hospital versus out-of-hospital pediatric cardiac arrest: a multicenter cohort study. Crit Care Med 2009;37:2259-67. [Crossref] [PubMed]
- Young KD, Seidel JS. Pediatric cardiopulmonary resuscitation: a collective review. Ann Emerg Med 1999;33:195-205. [Crossref] [PubMed]
- Meert KL, Donaldson A, Nadkarni V, et al. Multicenter cohort study of in-hospital pediatric cardiac arrest. Pediatr Crit Care Med 2009;10:544-53. [Crossref] [PubMed]
- Sandroni C, Ferro G, Santangelo S, et al. In-hospital cardiac arrest: survival depends mainly on the effectiveness of the emergency response. Resuscitation 2004;62:291-7. [Crossref] [PubMed]
- Skrifvars MB, Saarinen K, Ikola K, et al. Improved survival after in-hospital cardiac arrest outside critical care areas. Acta Anaesthesiol Scand 2005;49:1534-9. [PubMed]
- Stewart C, Shoemaker J, Keller-Smith R, et al. Code Team Training: Demonstrating Adherence to AHA Guidelines During Pediatric Code Blue Activations. Pediatr Emerg Care 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Berg RA, Sutton RM, Holubkov R, et al. Ratio of PICU versus ward cardiopulmonary resuscitation events is increasing. Crit Care Med 2013;41:2292-7. [Crossref] [PubMed]
- Kltcjmdm S. editor. To err is human: building a safer health system. Washington DC: National Academy Press, Institute of Medicine, 1999.
- Buist MD, Moore GE, Bernard SA, et al. Effects of a medical emergency team on reduction of incidence of and mortality from unexpected cardiac arrests in hospital: preliminary study. BMJ 2002;324:387-90. [Crossref] [PubMed]
- DeVita MA, Braithwaite RS, Mahidhara R, et al. Use of medical emergency team responses to reduce hospital cardiopulmonary arrests. Qual Saf Health Care 2004;13:251-4. [Crossref] [PubMed]
- Tibballs J, Kinney S, Duke T, et al. Reduction of paediatric in-patient cardiac arrest and death with a medical emergency team: preliminary results. Arch Dis Child 2005;90:1148-52. [Crossref] [PubMed]
- Brilli RJ, Gibson R, Luria JW, et al. Implementation of a medical emergency team in a large pediatric teaching hospital prevents respiratory and cardiopulmonary arrests outside the intensive care unit. Pediatr Crit Care Med 2007;8:236-46. [Crossref] [PubMed]
- Sharek PJ, Parast LM, Leong K, et al. Effect of a rapid response team on hospital-wide mortality and code rates outside the ICU in a Children's Hospital. JAMA 2007;298:2267-74. [Crossref] [PubMed]
- Hanson CC, Randolph GD, Erickson JA, et al. A reduction in cardiac arrests and duration of clinical instability after implementation of a paediatric rapid response system. Postgrad Med J 2010;86:314-8. [Crossref] [PubMed]
- Kotsakis A, Lobos AT, Parshuram C, et al. Implementation of a multicenter rapid response system in pediatric academic hospitals is effective. Pediatrics 2011;128:72-8. [Crossref] [PubMed]
- Kutty S, Jones PG, Karels Q, et al. Association of Pediatric Medical Emergency Teams With Hospital Mortality. Circulation 2018;137:38-46. [Crossref] [PubMed]
- Sen AI, Morgan RW, Morris MC. Variability in the implementation of rapid response teams at academic American pediatric hospitals. J Pediatr 2013;163:1772-4. [Crossref] [PubMed]
- McKelvie B, McNally JD, Chan J, et al. Increased Mortality and Length of Stay Associated With Medical Emergency Team Review in Hospitalized Pediatric Patients: A Retrospective Cohort Study. Pediatr Crit Care Med 2017;18:571-9. [Crossref] [PubMed]
- Jayaram N, Chan ML, Tang F, et al. Frequency of medical emergency team activation prior to pediatric cardiopulmonary resuscitation. Resuscitation 2017;115:110-5. [Crossref] [PubMed]
- Parshuram CS, Duncan HP, Joffe AR, et al. Multicentre validation of the bedside paediatric early warning system score: a severity of illness score to detect evolving critical illness in hospitalised children. Crit Care 2011;15:R184. [Crossref] [PubMed]
- Fenix JB, Gillespie CW, Levin A, et al. Comparison of Pediatric Early Warning Score to Physician Opinion for Deteriorating Patients. Hosp Pediatr 2015;5:474-9. [Crossref] [PubMed]
- Dean NP, Fenix JB, Spaeder M, et al. Evaluation of a Pediatric Early Warning Score Across Different Subspecialty Patients. Pediatr Crit Care Med 2017;18:655-60. [Crossref] [PubMed]
- Agulnik A, Forbes PW, Stenquist N, et al. Validation of a Pediatric Early Warning Score in Hospitalized Pediatric Oncology and Hematopoietic Stem Cell Transplant Patients. Pediatr Crit Care Med 2016;17:e146-53. [Crossref] [PubMed]
- Lambert V, Matthews A, MacDonell R, et al. Paediatric early warning systems for detecting and responding to clinical deterioration in children: a systematic review. BMJ Open 2017;7. [Crossref] [PubMed]
- Parshuram CS, Dryden-Palmer K, Farrell C, et al. Effect of a Pediatric Early Warning System on All-Cause Mortality in Hospitalized Pediatric Patients: The EPOCH Randomized Clinical Trial. JAMA 2018;319:1002-12. [Crossref] [PubMed]
- Rothman MJ, Tepas JJ 3rd, Nowalk AJ, et al. Development and validation of a continuously age-adjusted measure of patient condition for hospitalized children using the electronic medical record. J Biomed Inform 2017;66:180-93. [Crossref] [PubMed]
- da Silva YS, Hamilton MF, Horvat C, et al. Evaluation of Electronic Medical Record Vital Sign Data Versus a Commercially Available Acuity Score in Predicting Need for Critical Intervention at a Tertiary Children's Hospital. Pediatr Crit Care Med 2015;16:644-51. [Crossref] [PubMed]
- Endsley MR. Measurement of Situation Awareness in Dynamic Systems. Human Factors 1995;37:65-84. [Crossref]
- Endsley MR. Toward a theory of situation awareness in dynamic systems. Human Factors 1995;37:32-64. [Crossref]
- Endsley MR, Garland DJ. Situation Awareness: Analysis and Measurement. Mahwah, NJ: Lawrence Eribaum Associates, 2000.
- Brady PW, Zix J, Brilli R, et al. Developing and evaluating the success of a family activated medical emergency team: a quality improvement report. BMJ Qual Saf 2015;24:203-11. [Crossref] [PubMed]
- McClain Smith M, Chumpia M, Wargo L, et al. Watcher Initiative Associated With Decrease in Failure to Rescue Events in Pediatric Population. Hosp Pediatr 2017;7:710-5. [Crossref] [PubMed]
- Bonafide CP, Roberts KE, Priestley MA, et al. Development of a pragmatic measure for evaluating and optimizing rapid response systems. Pediatrics 2012;129:e874-81. [Crossref] [PubMed]
- Rubin J, Potes C, Xu-Wilson M, et al. An ensemble boosting model for predicting transfer to the pediatric intensive care unit. Int J Med Inform 2018;112:15-20. [Crossref] [PubMed]
- Wellner B, Grand J, Canzone E, et al. Predicting Unplanned Transfers to the Intensive Care Unit: A Machine Learning Approach Leveraging Diverse Clinical Elements. JMIR Med Inform 2017;5. [Crossref] [PubMed]


