
A learning curve of a novel multimodal endotracheal intubation assistant device for novices in a simulated airway: a prospective manikin trial with cumulative sum method
Introduction
Awake fiberoptic intubation is regularly performed in patients with anticipated difficult airways (1). However, many challenges are still associated with endotracheal intubation under the guidance of fiberoptic bronchoscopy. The visual field is limited and easily obstructed by secretions and blood in the airways (2). At the same time, learning to perform awake fiberoptic intubation is difficult and time-consuming for novice anesthesiologists (3). It is considered that more than 25 operations are needed for an anesthesiologist to be proficient with fiberoptic bronchoscopy (2). End-tidal carbon dioxide (ETCO2) is considered to facilitate endotracheal intubation with spontaneous breath (4,5). Previous studies have shown that ETCO2 monitoring assisted orientation and improved the safety of awake blind nasotracheal intubation (6,7) and reduced intubation time for inexperienced anesthesiologists (8). At present, there are still no intubation devices that integrate synchronous ETCO2 monitoring with visualization technology for clinical practice.
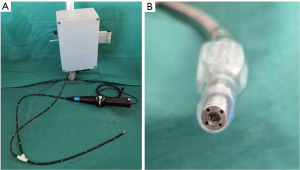
The multimodal endotracheal intubation assistant device (MEIAD, patent number: ZL202030298413.9) is a novel flexible intubation scope independently developed by the anesthesiology department at Shanghai Ninth People’s Hospital of Shanghai Jiao Tong University. The MEIAD prototype (Figure 1A) consisted of an operating handle, a guiding scope, and a sensor system. At the tip of the steerable, flexible scope, it was equipped with a camera and 4 gas sampling tubes (Figure 1B), which collected ETCO2 concentration from 4 directions based on the principle of infrared light absorption and used an advanced algorithm to locate the airway direction according to differences in ETCO2 concentration. The direction of the airway based on ETCO2 concentration was displayed on the screen simultaneously with the endoscopic image to assist operators in identifying airways during awake intubation, especially when the field of view is unclear.
The acquisition of the skill is also an important part of evaluating the utility of a device. The cumulative sum control chart (CUSUM) is a statistical analysis technique that can be used to evaluate learning curves (9). In medicine, CUSUM analysis can be used to monitor clinical performance and quality and to assess the time for operators to achieve expected levels (10). CUSUM is used in the learning analysis of many anesthesia practices, including intraspinal anesthesia, endotracheal intubation, and nerve blocks under the guidance of ultrasound (11-14). CUSUM is an efficient method to judge the number of attempts to achieve proficiency.
The objective of this study was to analyze the learning curve of MEIAD for inexperienced anesthesiology residents on a manikin by CUSUM analysis and evaluate the utility of the novel device. We present the following article in accordance with the STROBE reporting checklist (available at https://tp.amegroups.com/article/view/10.21037/tp-22-405/rc).
Methods
Participants
We conducted the prospective, observational, manikin trial from October 2020 to November 2020. The Clinical Research Ethics Committee of our institution deemed this study to be exempt from ethical review as it involved only a manikin and simulation training, which was considered a regular part of medical education. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013).
A total of 16 anesthesiology residents with 1–2 years clinical experience in our institution were enrolled. All participants had experience in nasal/orotracheal intubation with direct/video laryngoscopy. We excluded residents who had experience in fiberoptic bronchoscopy from our study. None of the residents had prior experience with MEIAD. All participants were voluntary and provided informed consent. The data collection was anonymous. The residents were named sequentially from A to P.
To simulate a difficult airway, we used a cervical collar to limit the mouth opening and neck extension of the manikin (BZ-51, Shanghai Baijiao, China). In addition, we connected the simulated lungs of the manikin to a CO2 accumulator, which generated CO2 when pressed to simulate exhaled CO2 during awake intubation (Figure 2).

Protocol and measurements
All participants received instruction and demonstration by an experienced anesthesiologist, including basic knowledge of airway anatomy and endotracheal intubation, an explanation of MEIAD, manipulation of the device, and demonstration on the manikin. Each resident then completed 40 exercises of nasotracheal insertion using MEIAD on the difficult airway manikin.
The manikin was positioned neutrally and the residents stood at the head of the manikin. After lubricating the guiding scope, the residents inserted the scope into the nasal cavity and slowly introduced the scope forward to find the glottis. An assistant pressed the CO2 accumulator at a frequency of 10 times per minute to simulate the expiration of CO2 in the airway. The residents adjusted the scope direction according to the indication of the airway analyzed by ETCO2 and the visualized image to see the glottis and then advanced the tip of the scope through the glottis until the carina was visible (Figure 3). The correct MEIAD positions were confirmed by the instructor viewing the image of the carina under the scope. In this study, the placement of an endotracheal tube was not required. We considered the insertion of the scope into the airway as the endpoint. In addition, the confidence in difficult airway management was scored subjectively by residents with a 11-point numerical rating scale (NRS) scale (ranging from 0 to 10, 0= very unconfident, 10= very confident) before and after 40 consecutive attempts.
The main data collected included success or failure and insertion time of each operation and the individual confidence score. Successful insertion of MEIAD was defined as correct placement of the device into the airway within 120 seconds. If the insertion time exceeded 120 seconds or if the scope entered into the esophagus, the operation was recorded as a failure. The insertion time was defined as the time from when the guiding scope entered the nasal cavity to when the tracheal carina was visible. The primary outcome was the learning curve of MEIAD established by CUSUM analysis. The secondary outcomes were an evaluation of the utility of MEIAD and the degree of improvement in confidence after simulation training.
Statistical analyses
To construct the learning curve according to CUSUM analysis, 4 basic variables were required: acceptable failure rate (p0), unacceptable failure rate (p1), standard type I error (α), and standard type II error (β) (15). Detailed calculations in CUSUM analysis based on the 4 variables are described in Table 1. The calculation of the CUSUM value started from zero. If a successful operation was performed, a value of S was subtracted from the previous CUSUM score. Conversely, a value of 1-S was added to the previous CUSUM score. Therefore, a negative trend in CUSUM values indicated a good trend for more successful attempts. When the curve was below the acceptable failure rate boundary h0, there was no significant difference between the actual and acceptable failure rate, and competence was considered obtained. If the CUSUM score was over the unacceptable failure rate boundary h1, the operator did not master the practice. When the curve score lines were between h1 and h0, it was not clear enough to judge the competence of operation.
Table 1
| Variables | Equations | Numeric values |
|---|---|---|
| α (type I error) | False positive | 0.1 |
| β (type II error) | False negative | 0.1 |
| p0 | Acceptable failure | 0.15 |
| p1 | Unacceptable failure | 0.3 |
| a | ln((1 − β)/α) | 2.19722457 |
| b | ln((1 − α)/β) | 2.19722457 |
| P | ln(p1/p0) | 0.693147181 |
| Q | ln((1 − p0)/(1 − p1)) | 0.194156014 |
| S (CUSUM) | Q/(P + Q) | 0.218815863 |
| h0 (Lower decision boundary) | −b/(P + Q) | −2.476295126 |
| h1 (Upper decision boundary) | a/(P + Q) | 2.476295126 |
| Number of cases for p0 | [(h0(1 − α) − αh1)/(s − p0)] | 36 |
| Number of cases for p1 | [(h1(1 − β) − βh0)/(p1 − s)] | 31 |
CUSUM, cumulative sum method.
As an exploratory study, 16 residents were included based on previous studies of learning curves and simulation training (16,17). The value of α and β were set conventionally as 0.1. In this study, we set p0 as 15% and p1 as 30% (p1 is usually twice the value of p0) (17). Based on the calculation, the number of operations required to achieve the acceptable failure rate p0 and unacceptable failure rate p1 was 36 and 31, respectively. Therefore, all residents attempted 40 insertions in the study.
The CUSUM chart of the learning curve was constructed by Microsoft Excel (Microsoft Office 2016) and statistical analyses were performed by SPSS 22.0 (IBM, Armonk, NY, USA). Continuous data are expressed by median (IQR, interquartile ranges), and categorical variables are expressed as percentages. We grouped the 40 insertions for every resident into 2 groups of 20 attempts each (phase 1: 1–20, phase 2: 21–40) to evaluate the learning effect of different periods. We used the Mann-Whitney test to compare insertion time and confidence score between the 2 groups because the data did not conform to a normal distribution. The success rate between periods was compared by the χ2 test. All tests were two-sided with a significance level of 0.05.
Results
All of the 16 novice residents (male/female: 8/8), aged 24.44±1.55, completed 40 trials of MEIAD (640 insertions in total), and all attempts were recorded completely. The CUSUM curves of the 16 novice residents using MEIAD are shown in Figure 4. According to the CUSUM analysis, 15 of 16 novice residents crossed the lower decision boundary h0 after 21.27±9.51 attempts. The 15 residents were considered proficient in endotracheal intubation with MEIAD on the difficult airway simulation to achieve an acceptable failure rate of 15%. Resident C had a CUSUM value between h0 and h1 after 40 attempts of MEIAD and was considered to not have obtained the skill at the acceptable level within 40 attempts.
The 40 insertions were stratified into 2 groups of 20 insertions each. The insertion success and insertion time of each group are shown in Table 2. The insertion time of the 2 groups was 24.0 (17.0–42.0) seconds vs. 17.5 (14.0–28.0) seconds, respectively (P<0.001). In terms of success rate, the difference between the 2 groups was also statistically significant (88.4% vs. 97.5%, respectively, P<0.001). As the number of attempts to master the use of MEIAD was approximately 22, it can be assumed that the last 20 attempts were the plateau of the learning curve. During the plateau period, the insertion time was 17.5 (14.0–28.0) seconds and the success rate was 97.5%.
Table 2
| Outcomes | Phase 1 | Phase 2 | P value |
|---|---|---|---|
| Insertion time(s) | 24.0 (17.0–42.0) | 17.5 (14.0–28.0) | <0.001 |
| Number of successes | 283 (88.4) | 312 (97.5) | <0.001 |
Data are shown as median (IQR) or number (%). MEIAD, multimodal endotracheal intubation assistant device; IQR, interquartile ranges.
In addition, after training on the simulation, the NRS score of the residents’ confidence in difficult airway management was significantly improved from 2.5 (1.3–4.0) to 7.0 (7.0–8.0).
Discussion
Our study showed that inexperienced residents achieved an accepted success rate of 15% using MEIAD in a difficult airway simulation after about 22 attempts. During the plateau period, the insertion time was 17.5 (14.0–28.0) seconds and the success rate was 97.5%.
With an incidence of 0.5–12.8%, the difficult airway is a major challenge for anesthesiologists during perioperative airway management (18). For anticipated difficult airway, awake endotracheal intubation under the guidance of fiberoptic bronchoscope is recommended to guarantee patients’ security. Some relevant studies have shown that the success rate of awake fiberoptic bronchoscope intubation was 78–100% (19-22). Although fiberoptic bronchoscopy is advised for difficult airways, it takes considerable training to obtain the skill, particularly for novice residents (23,24). In addition, fiberoptic bronchoscope is easily affected by sputum and secretions from the oropharynx or nasopharynx, making the glottis difficult to identify. The novel device added indication of airway based on the algorithm of ETCO2 to the endoscope in order to play an auxiliary role in difficult airway awake intubation with unclear vision or abnormal anatomy. The first-generation prototype of the instrument was completed and feasibility of endotracheal intubation was preliminarily verified on pigs with spontaneous breath in our previous study (25). In this study, we focused on the learning curve and efficacy of the novel device.
As a novel device, it is similar to a fiberscope except for the multimodal indication of airway direction. For acquisition of the awake intubation skill with fiberscope, Smith et al. observed that after performing 18 fiberoptic nasotracheal intubations in patients with normal airways, trainees could complete 70–80% intubations within 1 minute, and it took about 45 manipulation attempts to reach the expected “expert level” (26). Dalal et al. analyzed the learning curve of 16 residents in a study using fiberscope in a normal airway simulation for nasotracheal intubations, and it took 27–58 attempts to be proficient in nasotracheal fiberoptic intubation (27). Moreover, once proficiency was attained, the average operation time of fiberoptic bronchoscopy was about 50–120 seconds in previous studies (28,29). The differences in learning curves, operation time, and success rate among the studies may be related to the airway simulations and varying definitions of success. In our study, the insertion time was defined from the entry of the nasal cavity to the vision of the carina. We didn’t perform subsequent endotracheal intubation and the insertion time was defined as the time of scope examination because the main purpose of this study was to explore the learning curve and utility of this novel intubation device. The process of advancing endotracheal tubes may have an impact on the utility and effectiveness of the device itself (30). In addition, the identification and positioning of airway is considered a crucial part of endotracheal intubation. Successful identification of the airway and delivery of the guiding scope into the airway have positive implications for the completion of endotracheal intubation operation. Therefore, in our study, the main endpoint was the correct insertion of the novel device instead of completion of the entire endotracheal intubation process. With the accumulation of experience, the differences in insertion time and success rate between the first 20 attempts and the last 20 attempts were statistically significant, and it was considered that the learning plateau period was reached at the last 20 attempts. Therefore, with acceptable training on the simulation, the insertion time of MEIAD on the manikin was 17.5 (14.0–28.0) seconds, and the success rate was 96.6%. As a novel and self-developed intubation assistant device, MEIAD showed a satisfactory learning curve and efficacy on the manikin.
A learning curve is a way to monitor performance development over time. There are many ways to construct a learning curve, such as with a graph, table, or statistical technique (31). CUSUM analysis is a useful method for spotting subtle, slow, prolonged degradation in a process under control and thus an effective method to evaluate a novel skill. However, CUSUM analysis requires a clear description of the success and failure of the operation, and the assessment condition cannot be changed randomly during the research process. In terms of endotracheal intubation, the success and failure of the operation are not uniformly defined. In early relevant studies involving CUSUM analysis of endotracheal intubation, the p0, p1, α, and β values were also set differently. The requirement for strict quality control in medical training is important, but there is still debate over whether or not these restrictions should be gentler for novice residents. Since this study was an exploratory study of a novel device prototype and the operators were novice anesthesiologists (grades 1–2), we set the acceptable failure rate at 15% and defined success as the correct insertion into the airway within 120 seconds. These variables were identical to prior CUSUM-based studies regarding learning curves of tracheal intubation (16,17). Under this condition, the residents became proficient at using MEIAD on the difficult airway simulator after 22 manipulations. If a stringent acceptable failure rate is set or success is defined as a shorter operation time, the number of people who acquire skills within the specified attempts may also decrease accordingly. For example, setting the acceptable failure rate as 10%, only 11 residents could cross h0 within 40 manipulation attempts. If a successful operation was defined as less than 90 seconds, 2 residents could not cross the lower decision boundary h0 in 40 operation attempts. Therefore, the results of CUSUM analysis of the learning curve need to be comprehensively considered in combination with the assigned p0, p1, α, and β values.
Simulation-based education is an important approach for teaching and training in difficult airway management (32,33). For some unconventional operations with a slow learning curve, simulation-based training can be especially effective for reducing harm to patients caused by novices in the early stage of clinical operations. Consistent with the results of our study, simulated practice is an effective approach for improving the self-confidence of beginners. Since MEIAD is still in the development and verification stage, a simulated airway is suitable for initial exploration.
There were several limitations to this study. Our study involved the use of a manikin, and due to its relatively simple structure, the results obtained on the simulator cannot be fully replicated in clinical practice under the same conditions (34). It is certain that the clinical process will be more challenging and the learning curve will decline more gradually. Further, according to the literature, common physical methods for constructing a difficult airway are cervical collar and simulated tongue edema (35). The advantage of MEIAD is to assist with an obscured or unclear view, and the simulated tongue edema method is more suitable for constructing an unclear view. Due to the limitation of the manikin, the tongue was unable to be inflated to simulate tongue edema. At the same time, it also cannot simulate the clinical scenario of sputum and secretions blocking the view field (36), so the advantage of multimodal tracheal intubation aids was not fully realized. The initial prototype MEIAD used in this study has not yet been put into production and clinical application. As a proof-of-concept study, we confirmed the utility and efficacy of MEIAD on the airway simulator, and further confirmation in a different clinical setting or involving operators with different experience is needed. Considering the diversity and complexity of actual clinical scenarios, the device will be modified and improved before clinical application.
In conclusion, novice residents could be basically proficient in MEIAD after 22 practice sessions on the simulated airway. At the stable stage of the training, the insertion time was 17.5 (14.0–28.0) seconds, and the success rate was 97.5%. It is expected that MEIAD will be further improved and promoted and has the potential to be an alternative airway device.
Acknowledgments
Funding: This study was supported by the Clinical Research Plan of the Shenkang Hospital Development Center (No. SHDC2020CR3043B) and Faculty Development Training Programme of the School of Medicine, Shanghai Jiao Tong University (No. JFXM202109).
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tp.amegroups.com/article/view/10.21037/tp-22-405/rc
Data Sharing Statement: Available at https://tp.amegroups.com/article/view/10.21037/tp-22-405/dss
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tp.amegroups.com/article/view/10.21037/tp-22-405/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The Clinical Research Ethics Committee of our institution deemed this study to be exempt from ethical review as it involved only a manikin and simulation training, which was considered a regular part of medical education. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). All participants were voluntary and provided informed consent.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Apfelbaum JL, Hagberg CA, Connis RT, et al. 2022 American Society of Anesthesiologists Practice Guidelines for Management of the Difficult Airway. Anesthesiology 2022;136:31-81. [Crossref] [PubMed]
- Moore A, Schricker T. Awake videolaryngoscopy versus fiberoptic bronchoscopy. Curr Opin Anaesthesiol 2019;32:764-8. [Crossref] [PubMed]
- Murphy T, Howes B. Current practice for awake fibreoptic intubation - some unanswered questions. Anaesthesia 2017;72:678-81. [Crossref] [PubMed]
- Li T, Liu T, Li M, et al. ETCO2 waveforms-assisted awake nasal fibreoptic intubation. J Clin Monit Comput 2021;35:1525-7. [Crossref] [PubMed]
- Lee LY, Lee D, Ryu H, et al. Capnography-guided Endotracheal Intubation as an Alternative to Existing Intubation Methods in Rabbits. J Am Assoc Lab Anim Sci 2019;58:240-5. [Crossref] [PubMed]
- King HK, Wooten JD. Blind nasal intubation by monitoring end-tidal CO2. Anesth Analg 1989;69:412-3. [Crossref] [PubMed]
- Dohi S, Inomata S, Tanaka M, et al. End-tidal carbon dioxide monitoring during awake blind nasotracheal intubation. J Clin Anesth 1990;2:415-9. [Crossref] [PubMed]
- Liu BY, Yan J, Jiang H. Application of end tidal carbon dioxide monitoring in difficult tracheal intubation during oral and maxillofacial surgery. China J Oral Maxillofac Surg 2019;17:448-52.
- Lew E, Allen JC Jr, Goy RWL, et al. Determining competence in performing obstetric combined spinal-epidural procedures in junior anesthesiology residents: results from a cumulative sum analysis. Int J Obstet Anesth 2020;44:33-9. [Crossref] [PubMed]
- Narayanasamy S, Ding L, Yang F, et al. Feasibility study of cumulative sum (CUSUM) analysis as a competency assessment tool for ultrasound-guided venous access procedures. Can J Anaesth 2022;69:256-64. [Crossref] [PubMed]
- Starkie T, Drake EJ. Assessment of procedural skills training and performance in anesthesia using cumulative sum analysis (cusum). Can J Anaesth 2013;60:1228-39. [Crossref] [PubMed]
- Oliveira KF, Arzola C, Ye XY, et al. Determining the amount of training needed for competency of anesthesia trainees in ultrasonographic identification of the cricothyroid membrane. BMC Anesthesiol 2017;17:74. [Crossref] [PubMed]
- Arnuntasupakul V, Chalachewa T, Leurcharusmee P, et al. Ultrasound with neurostimulation compared with ultrasound guidance alone for lumbar plexus block: A randomised single blinded equivalence trial. Eur J Anaesthesiol 2018;35:224-30. [Crossref] [PubMed]
- Orhan-Sungur M, Altun D, Özkan-Seyhan T, et al. Learning curve of ultrasound measurement of subglottic diameter for endotracheal tube selection in pediatric patients. Paediatr Anaesth 2019;29:1194-200. [Crossref] [PubMed]
- Chuan A, Wan AS, Royse CF, et al. Competency-based assessment tools for regional anaesthesia: a narrative review. Br J Anaesth 2018;120:264-73. [Crossref] [PubMed]
- Nakanishi T, Sakamoto S, Yoshimura M, Toriumi T. A learning curve of LMA® ProSeal™ insertion: a prospective analysis of cumulative sum method. J Anesth 2020;34:554-60. [Crossref] [PubMed]
- Altun D, Ozkan-Seyhan T, Camci E, et al. Learning Curves for Two Fiberscopes in Simulated Difficult Airway Scenario With Cumulative Sum Method. Simul Healthc 2019;14:163-8. [Crossref] [PubMed]
- Edelman DA, Perkins EJ, Brewster DJ. Difficult airway management algorithms: a directed review. Anaesthesia 2019;74:1175-85. [Crossref] [PubMed]
- Aziz MF, Brambrink AM, Healy DW, et al. Success of Intubation Rescue Techniques after Failed Direct Laryngoscopy in Adults: A Retrospective Comparative Analysis from the Multicenter Perioperative Outcomes Group. Anesthesiology 2016;125:656-66. [Crossref] [PubMed]
- Cabrini L, Baiardo Redaelli M, Ball L, et al. Awake Fiberoptic Intubation Protocols in the Operating Room for Anticipated Difficult Airway: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Anesth Analg 2019;128:971-80. [Crossref] [PubMed]
- El-Boghdadly K, Onwochei DN, Cuddihy J, et al. A prospective cohort study of awake fibreoptic intubation practice at a tertiary centre. Anaesthesia 2017;72:694-703. [Crossref] [PubMed]
- Wahdan AS, El-Refai NAR, Omar SH, et al. Endotracheal intubation in patients undergoing open abdominal surgery in the lateral position: a comparison between the intubating video stylet and fiberoptic intubating bronchoscopy. Korean J Anesthesiol 2021;74:234-41. [Crossref] [PubMed]
- Ovassapian A, Yelich SJ, Dykes MH, et al. Learning fibreoptic intubation: use of simulators v. traditional teaching. Br J Anaesth 1988;61:217-20. [Crossref] [PubMed]
- Desai N, Ratnayake G, Onwochei DN, et al. Airway devices for awake tracheal intubation in adults: a systematic review and network meta-analysis. Br J Anaesth 2021;127:636-47. [Crossref] [PubMed]
- Xia M, Xu T, Cao S, et al. Evaluation of a Novel Multimodal Guidance Device for Difficult Airway Endotracheal Intubation in Spontaneously Breathing Pigs. Journal of Shanghai Jiaotong University (Science) 2022;27:256-63. [Crossref]
- Smith JE, Jackson AP, Hurdley J, et al. Learning curves for fibreoptic nasotracheal intubation when using the endoscopic video camera. Anaesthesia 1997;52:101-6. [Crossref] [PubMed]
- Dalal PG, Dalal GB, Pott L, et al. Learning curves of novice anesthesiology residents performing simulated fibreoptic upper airway endoscopy. Can J Anaesth 2011;58:802-9. [Crossref] [PubMed]
- László CJ, Szűcs Z, Nemeskéri Á, et al. Human cadavers preserved using Thiel's method for the teaching of fibreoptically-guided intubation of the trachea: a laboratory investigation. Anaesthesia 2018;73:65-70. [Crossref] [PubMed]
- Nilsson PM, Russell L, Ringsted C, et al. Simulation-based training in flexible fibreoptic intubation: A randomised study. Eur J Anaesthesiol 2015;32:609-14. [Crossref] [PubMed]
- Biro P, Hofmann P, Gage D, et al. Automated tracheal intubation in an airway manikin using a robotic endoscope: a proof of concept study. Anaesthesia 2020;75:881-6. [Crossref] [PubMed]
- Howard NM, Cook DA, Hatala R, et al. Learning Curves in Health Professions Education Simulation Research: A Systematic Review. Simul Healthc 2021;16:128-35. [Crossref] [PubMed]
- Grande B, Kolbe M, Biro P. Difficult airway management and training: simulation, communication, and feedback. Curr Opin Anaesthesiol 2017;30:743-7. [Crossref] [PubMed]
- Chen Q, Hu Z, Wang Q, et al. Application of a Simulator-Based Teaching Method in the Training of the Flexible Bronchoscope-Guided Intubation. Curr Health Sci J 2021;47:501-6. [PubMed]
- Baciarello M, Zasa M, Manferdini ME, et al. The learning curve for laryngoscopy: Airtraq versus Macintosh laryngoscopes. J Anesth 2012;26:516-24. [Crossref] [PubMed]
- Aghamohammadi H, Massoudi N, Fathi M, et al. Intubation learning curve: comparison between video and direct laryngoscopy by inexperienced students. J Med Life 2015;8:150-3. [PubMed]
- Rhee SH, Yun HJ, Kim J, et al. Risk factors affecting the difficulty of fiberoptic nasotracheal intubation. J Dent Anesth Pain Med 2020;20:293-301. [Crossref] [PubMed]



