Recurrent wheezing in children
Introduction
Wheezing during early life represents a common disorder characterized by airways obstruction (1).
Recurrent wheezing have a significant morbidity and it’s estimated that about one third of school-age children manifest the symptom during the first 5 years of life (2).
In young children, wheezing, either transient or persistent, can be severe and cause a poor quality of life with frequent use of health care system and economic costs (1,2).
Wheezing is a common problem worldwide and the most frequent causes of wheezing in preschool children are bronchiolitis and asthma (1,3).
Parents of infants with recurrent wheezing often ask the pediatrician: “Will my children develop asthma?” This is a crucial question that involved also clinician in the diagnostic and therapeutic at tempts.
Proper identification of children at risk of developing asthma at school age may predict long-term outcomes and improve treatment and preventive approach, but the possibility to identify these children at preschool age remains limited.
Wheezing is a multi-factorial symptom, usually related to bronchiolitis or asthma, but other less common conditions may be consider in case of atypical presentation (1). Although most of the patients (60%) are expected to improve and to be symptom-free at the age of 6 years and the majority of them remain asymptomatic at the age of 11 and 16 years (4).
To better define the patients with wheezing, a tempts of classification, identification of asthma risk factors, genetic and environmental factors has been proposed to improve the characterization of children with recurrent wheezing.
Differential diagnosis
The differential diagnosis of chronic or recurrent wheezing is broad and includes structural or non-structural causes (Table 1).

Full table
Structural defects include anatomic abnormalities such as tracheobronchial tree malacia and vascular ring or slings that manifest themselves early in life, typically in the first few months and are not responding to the therapy.
Non-structural causes are represented by various conditions that are related with recurrent wheezing. The most common ones are aspiration syndrome, foreign body inhalation, gastro esophageal reflux, fistulas and swallowing disorders related to neurologic or muscular dysfunction (5).
Recurrent infections of the lower respiratory tract can present as recurrent wheezing and host defense abnormalities may be considered.
Cystic fibrosis, bronchopulmonary dysplasia, obliterans bronchiolitis, interstitial lung disease and paradoxical vocal cord dysfunction are other causes to recognize.
Wheezing phenotypes
For many years authors focused their studies to identify early children with recurrent wheezing at risk to develop asthma at school age. They have proposed several phenotypes for a more precise characterization and a personalized plan of treatment. In 1995, Martinez et al., for the first time, introduced a classification focusing on three phenotypes: “transient wheezing”, “persistent wheezing” and “late onset wheezing” focusing on temporal appearance and persistence of symptoms (6). In 2008, an ERS task force reassessed the topic focusing on frequency and duration of associated symptoms and triggers (7). In particular wheezing in children were classified as “episodic viral wheezing” (EVW) or “multiple trigger wheezing” (MTW).
Recently, some studies revised the previous classifications of wheezing since they failed to be useful in clinical practice to manage children who were classified on symptom frequency and severity (8).
The main criticism concerns the inability to define stable phenotypes with the risk of overestimating or underestimating the characteristics of symptoms in these children (9).
In particular some authors of the 2008 ERS task force revised the contents taking into account the scientific evidence and clinical practice considerations (9).
The new document underlines how the previous classification, including EVW and MTW, was not useful and applicable in clinical practise because symptoms in children can be variable during the time and the two phenotypes can sometimes overlap (9).
Furthermore GINA guidelines revised in 2014 highlight the concept that asthma is already manifest in children under 5 years (10).
In this document authors emphasize the difficulty to diagnosis asthma at this age because of the frequency of episodes of upper respiratory infection and the lack of diagnostic instruments as spirometry to define bronchial-reversibility.
For this purpose GINA guidelines proposed frequency, gravity and duration of symptoms, inter critical manifestations, family history of atopy as tools to guide clinicians in asthma diagnosis and therapy (10).
It is suggested that a child with two or three episodes of wheezing in a year, lasting less 10 days, without symptoms between episodes has probably a viral bronchospasm with low probability to develop asthma (10). On the other hand a child with recurrent and longer episodes, familiar history of atopy associated with exercise induced wheezing has a higher probability to have asthma.
In the GINA document frequency, gravity, duration and time course of symptoms are recognized as the key features that the clinician must follow in diagnostic and therapeutic decision.
Asthma risk factors
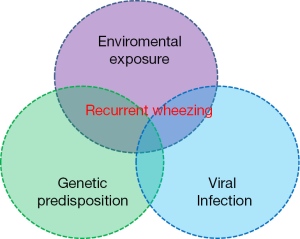
A number of studies demonstrated that asthma is a multi-factorial disease results from the interaction between genetic and environmental factors (Figure 1) (11).
Genetic studies on asthma revealed a large number of candidate genes associated with immune system that are potentially involved in pathogenesis of the disease (12).
Yang et al showed that cord blood IgE level, male sex, second hand cigarette smoke and parental history of atopy are predictive risk factors for recurrent wheezing (13). In particular they found a correlation with Clara cell protein CC10 G+38A polymorphism and lower CC10 levels in children with recurrent wheezing (13).
Other genetic markers were investigated, such as IL33-IL1RL1 pathway that has been associated with intermediate, late onset and persistent wheeze (14).
Other studies were performed about genetic involvement in asthma as a polymorphism in TNF-α as a potential genetic factor contributing in the development of wheezing and asthma (15).
Although further studies are still needed these findings may be useful for the early identification of children at the highest risk of developing recurrent episodes and the possibility to develop a specific pharmacotherapy.
Some studies have evaluated the relationship between atopy and asthma. In Tucson Children’s Respiratory Study Cohort (TCRS) predisposition to allergy appeared to be a primary risk factor in children with wheezing (6). Patients with late onset and recurrent wheezing had a higher sensitization to allergen at 6 ages those children without wheezing.
Children with IgE associated atopic persistent phenotype have the symptom that persists into adolescence (6). On the contrary children with non-atopic persistent wheezing generally have the first episode during first years of life and the wheezing episodes became less frequent in adolescence (16).
Environmental factors are also involved in asthma develop and exacerbations.
The hygiene hypothesis proposes that fewer infections in early life and lower exposure to microbes may shift the immune system from Th1 to Th2-biased allergic response in early life (17).
Changes in the intestinal microbiome have been demonstrated to be also involved in the increase of asthma prevalence and other allergic conditions (18).
Exposure to tobacco smoke, crowding, pets, dust mite are all considered important indoor allergens and irritants (18).
Air pollution, also, causes asthma exacerbations and increases hospital admission for respiratory symptoms (19).
Tobacco smoke is an important indoor air pollutant that is involved in asthma development during childhood (20). As early is fetal exposure to maternal smoking during pregnancy the risk increases. During intrauterine exposure tobacco by products are transferred from placenta to the foetus (20). In an animal model epigenetic changes due to tobacco exposure during pregnancy increased the risk of wheezing therefore suggesting asthma risk after birth (21).
Recently has been proposed a role of vitamin D and antioxidant on development of asthma. The effect of vitamin D and trace elements has been discussed in recurrent wheezing. In particular there is a positive correlation between a decreased intake of antioxidants and increased incidence of the disease (22).
Vitamin D and trace elements can act as anti-inflammatory and anti-oxidant agents on host immune system. A reduction of these elements in diet intake increases oxidant damage and derangement of the immune system (23).
A diet poor in antioxidants makes the host more vulnerable to reactive oxygen species. Vitamin D and trace elements have an immune modulator effect and can be effective in respiratory tract infections.
Predictive index in children with wheezing
In the last years there has been a significant increase of asthma diagnosis in children and a decrease in the age of asthma diagnosis (24).
Various asthma predictive indexes (APIs) have been developed in the last years to identify children at risk of asthma in preschool age. Epidemiologic studies have employed various risk factors associated with the development of asthma such as parental history of atopy, wheezing history, IgE levels and cytokines profiles.
API is a validated clinical model for childhood asthma defined on a cohort of children who wheeze at least one time during the first 3 years of life (25-27).
The primary criteria to identify the score are ≥4 episodes of wheezing in 1 year and the secondary are clinician diagnose of parental eczema or asthma, allergic sensitization to aeroallergen, wheezing unrelated to cold, eosinophilia ≥4 percent (26). A positive index was defined as at least major criterion plus at least one major or two minor criteria (26).
API sensitivity is low, suggesting that the test is poor for predicting later asthma development. Nevertheless API has a high negative predictive value, meaning that it can identify children who have a low probability to develop asthma with a negative test.
A modified version (mAPI) was tested in a cohort of high risk children with a family history of allergy and/or asthma (28). A positive mAPI increased the probability to identify patient at risk to develop future asthma.
Another simple tool was developed by Pescatore et al. (29). The questionnaire is a simple and robust tool to assess the prevalence of asthma 5 years later in preschool children with wheeze or cough (29).
The index considers only non-invasive predictors that are easy to assess in primary care such as demographic and perinatal data, eczema, upper and lower respiratory tract symptoms and family history of atopy. The score can stratify patients with high, medium or low risk to develop asthma. This is a simple, low-cost and noninvasive method to predict the risk of later asthma in symptomatic preschool children.
Therapeutic strategy
The approach to the treatment of preschool wheezing has been widely discussed and recently revised (9,10). A significant reconsideration of the role of ICS in this population has been based on a review and meta-analysis considering 29 studies in pre-schooler wheezers (30).
The meta-analysis has demonstrated that ICS are effective in reducing the frequency of wheezing episodes during preschool age, irrespective of phenotype or atopy status (30).
Furthermore, authors underline the importance of the adherence to ICS therapy in children aged two-six years in the control of asthma symptoms.
The re-emerging potential role of ICS in the treatment of wheezing in preschool age has also opened the question whether the preferred treatment in this categories should be low dose ICS daily or higher dose ICS intermittent.
A couple of studies showed no difference in number and severity of exacerbations between the two strategies with a lower ICS use with the intermittent design.
The intermittent use of high dose of ICS represents one a relevant new option in the management of wheezing in pre-schooler proposed by either 2014 ERJ review and GINA document (9,10).
This therapeutic strategy was proposed by a number of studies show in the last few years.
Bacharier et al. evaluated the efficacy of high intermittent dose of budesonide (1,000 mcg twice daily) for one week when the patient has the first symptoms of respiratory infection in preschool children with recurrent wheezing. They demonstrated a significative reduction in severity of relapses in particular in children with a positive API.
The BEST study showed that a regular low dose of BPD was the most effective treatment for preschool children with recurrent wheezing. A valid alternative was intermittent salbutamol and BPD (80 mcg daily) during exacerbations with a significant reduction in ICS total dose (31).
Ducharme et al. showed also a reduction in the use of oral corticosteroid in preschool children with the intermitted high dose use of fluticasone (1,500 mcg/day), but it underlines the warning on a negative effect on growth in a half of patients at such high dosage (32). The dose utilised in the study in fact is far higher than usually use in clinical practice.
A study evaluated the effect of high dose of nebulized BPD in children with viral wheeze (33). The authors observed a reduction in the number of episodes and number of children with wheeze in the group with active drug compared with placebo.
These studies support the “non-genomic effect” of ICS in the treatment of episodic wheezing in preschool children. In fact while the anti-inflammatory effect of ICS is based on the cellular activation by genomic pathways the “non-genomic” effect based on vasoconstriction and reduction of airways blood flow, relevant during preschool wheezing due to viral infections (34).
The two most significant studies on intermittent ICS use were performed using pneumatic nebulizer (31,35). This issue is very relevant in the real life setting. In fact, although guidelines consider metered dose inhalers (MDI) plus spacer as first choice, in the real life nebulizers are commonly used in both USA and Europe (10).
These results also support the recommendation from the group of ERS experts and the recent revision of GINA document. In particular, GINA initiative has a dedicate chapter on diagnosis and management of asthma in children under five. The approach proposed in GINA document is focused on frequency and severity of wheezing episodes in correlation with inter critical symptoms and family history of atopy in order to define the risk of children to develop asthma. A child with two or three episodes in 1 year, each lasting less than 10 days without inter critical symptoms has less probability to be asthmatic and to have benefit from a regular controller therapy. On the other hand, a child with frequent, long lasting symptoms, exercise induced wheezing and a family history of atopy is more probably an asthmatic subject for whom a low regular dose of ICS therapy may be consider the most appropriate treatment.
Furthermore, intermitted ICS was also proposed for children with intermittent viral induced wheeze without symptoms between episodes, if short acting β2 agonist (SABA) is not sufficient.
Conclusions
In the last years wheezing in preschool children has been widely reconsidered.
The classification of children with wheezing is now mainly based upon the severity and frequency of episodes rather on specific phenotypes, previously proposed.
The therapeutic approach is now based upon symptoms severity and frequency. A regular ICS treatment is consider more effective in asthmatic children and in those with recurrent wheezing and inter critical symptoms. Intermittent ICS is now considered as a valid option for preschool children with intermittent viral induced wheezing and no interval symptoms.
Acknowledgements
None.
Footnote
Conflicts of Interest: G Piacentini has served in advisory boards for Aerocrine AB, Chiesi S.p.A., Sensor Medics Italia s.r.l., Stallergenes Italia s.r.l. and received lectures fees for Aerocrine AB, Chiesi S.p.A., Italchimici S.p.A., Meda Pharma S.p.A., ThermoFisher/Phadia s.r.l. The other authors have no conflicts of interest to declare.
References
- Ducharme FM, Tse SM, Chauhan B. Diagnosis, management, and prognosis of preschool wheeze. Lancet 2014;383:1593-604. [PubMed]
- Mallol J, García-Marcos L, Solé D, et al. International prevalence of recurrent wheezing during the first year of life: variability, treatment patterns and use of health resources. Thorax 2010;65:1004-9. [PubMed]
- Garcia-Marcos L, Mallol J, Solé D, et al. International study of wheezing in infants: risk factors in affluent and non-affluent countries during the first year of life. Pediatr Allergy Immunol 2010;21:878-88. [PubMed]
- Martinez FD. What have we learned from the Tucson Children's Respiratory Study? Paediatr Respir Rev 2002;3:193-7. [PubMed]
- Celedón JC, Litonjua AA, Ryan L, et al. Bottle feeding in the bed or crib before sleep time and wheezing in early childhood. Pediatrics 2002;110:e77. [PubMed]
- Martinez FD, Wright AL, Taussig LM, et al. Asthma and wheezing in the first six years of life. The Group Health Medical Associates. N Engl J Med 1995;332:133-8. [PubMed]
- Brand PL, Baraldi E, Bisgaard H, et al. Definition, assessment and treatment of wheezing disorders in preschool children: an evidence-based approach. Eur Respir J 2008;32:1096-110. [PubMed]
- Savenije OE, Kerkhof M, Koppelman GH, et al. Predicting who will have asthma at school age among preschool children. J Allergy Clin Immunol 2012;130:325-31. [PubMed]
- Brand PL, Caudri D, Eber E, et al. Classification and pharmacological treatment of preschool wheezing: changes since 2008. Eur Respir J 2014;43:1172-7. [PubMed]
- Global iniziative for Asthma. Global strategy for asthma management and prevention. Available online: http://www.ginasthma.org/local/uploads/files/GINA_Report2015_Tracked.pdf
- Lasso-Pirot A, Delgado-Villalta S, Spanier AJ. Early childhood wheezers: identifying asthma in later life. J Asthma Allergy 2015;8:63-73. [PubMed]
- Renkonen J, Joenväärä S, Parviainen V, et al. Network analysis of single nucleotide polymorphisms in asthma. J Asthma Allergy 2010;3:177-86. [PubMed]
- Yang KD, Ou CY, Chang JC, et al. Infant frequent wheezing correlated to Clara cell protein 10 (CC10) polymorphism and concentration, but not allergy sensitization, in a perinatal cohort study. J Allergy Clin Immunol 2007;120:842-8. [PubMed]
- Savenije OE, Mahachie John JM, Granell R, et al. Association of IL33-IL-1 receptor-like 1 (IL1RL1) pathway polymorphisms with wheezing phenotypes and asthma in childhood. J Allergy Clin Immunol 2014;134:170-7. [PubMed]
- Shaker OG, Sadik NA, El-Hamid NA. Impact of single nucleotide polymorphism in tumor necrosis factor-α gene 308G/A in Egyptian asthmatic children and wheezing infants. Hum Immunol 2013;74:796-802. [PubMed]
- Stein RT, Sherrill D, Morgan WJ, et al. Respiratory syncytial virus in early life and risk of wheeze and allergy by age 13 years. Lancet 1999;354:541-5. [PubMed]
- Le Cann P, Bonvallot N, Glorennec P, et al. Indoor environment and children's health: recent developments in chemical, biological, physical and social aspects. Int J Hyg Environ Health 2011;215:1-18. [PubMed]
- Kanchongkittiphon W, Gaffin JM, Phipatanakul W. The indoor environment and inner-city childhood asthma. Asian Pac J Allergy Immunol 2014;32:103-10. [PubMed]
- Heroux ME, Braubach M, Korol N, et al. The main conclusions about the medical aspects of air pollution: the projects REVIHAAP and HRAPIE WHO/EC. Gig Sanit 2013.9-14. [PubMed]
- Gilmour MI, Jaakkola MS, London SJ, et al. How exposure to environmental tobacco smoke, outdoor air pollutants, and increased pollen burdens influences the incidence of asthma. Environ Health Perspect 2006;114:627-33. [PubMed]
- Magnus MC, Håberg SE, Karlstad Ø, et al. Grandmother's smoking when pregnant with the mother and asthma in the grandchild: the Norwegian Mother and Child Cohort Study. Thorax 2015;70:237-43. [PubMed]
- van Oeffelen AA, Bekkers MB, Smit HA, et al. Serum micronutrient concentrations and childhood asthma: the PIAMA birth cohort study. Pediatr Allergy Immunol 2011;22:784-93. [PubMed]
- Soutar A, Seaton A, Brown K. Bronchial reactivity and dietary antioxidants. Thorax 1997;52:166-70. [PubMed]
- Radhakrishnan DK, Dell SD, Guttmann A, et al. Trends in the age of diagnosis of childhood asthma. J Allergy Clin Immunol 2014;134:1057-62.e5.
- Castro-Rodríguez JA, Holberg CJ, Wright AL, et al. A clinical index to define risk of asthma in young children with recurrent wheezing. Am J Respir Crit Care Med 2000;162:1403-6. [PubMed]
- Castro-Rodriguez JA. The Asthma Predictive Index: early diagnosis of asthma. Curr Opin Allergy Clin Immunol 2011;11:157-61. [PubMed]
- Leonardi NA, Spycher BD, Strippoli MP, et al. Validation of the Asthma Predictive Index and comparison with simpler clinical prediction rules. J Allergy Clin Immunol 2011;127:1466-72.e6.
- Amin P, Levin L, Epstein T, et al. Optimum predictors of childhood asthma: persistent wheeze or the Asthma Predictive Index? J Allergy Clin Immunol Pract 2014;2:709-15. [PubMed]
- Pescatore AM, Dogaru CM, Duembgen L, et al. A simple asthma prediction tool for preschool children with wheeze or cough. J Allergy Clin Immunol 2014;133:111-8.e1-13.
- Castro-Rodriguez JA, Rodrigo GJ. Efficacy of inhaled corticosteroids in infants and preschoolers with recurrent wheezing and asthma: a systematic review with meta-analysis. Pediatrics 2009;123:e519-25. [PubMed]
- Papi A, Nicolini G, Baraldi E, et al. Regular vs prn nebulized treatment in wheeze preschool children. Allergy 2009;64:1463-71. [PubMed]
- Ducharme FM, Lemire C, Noya FJ, et al. Preemptive use of high-dose fluticasone for virus-induced wheezing in young children. N Engl J Med 2009;360:339-53. [PubMed]
- Ghirga G, Ghirga P, Fagioli S, et al. Intermittent treatment with high dose nebulized beclomethasone for recurrent wheezing in infants due to upper respiratory tract infection. Minerva Pediatr 2002;54:217-20. [PubMed]
- Wanner A, Horvath G, Brieva JL, et al. Nongenomic actions of glucocorticosteroids on the airway vasculature in asthma. Proc Am Thorac Soc 2004;1:235-8. [PubMed]
- Zeiger RS, Mauger D, Bacharier LB, et al. Daily or intermittent budesonide in preschool children with recurrent wheezing. N Engl J Med 2011;365:1990-2001. [PubMed]