
Autism spectrum disorder: definition, epidemiology, causes, and clinical evaluation
Definition
Autism spectrum disorder (ASD) is a neurodevelopmental disorder characterized by deficits in social communication and the presence of restricted interests and repetitive behaviors (1). In 2013, the Diagnostic and Statistical Manual of Mental Disorders—5th edition (DSM-5) was published, updating the diagnostic criteria for ASD from the previous 4th edition (DSM-IV) (Table 1) (1,2).
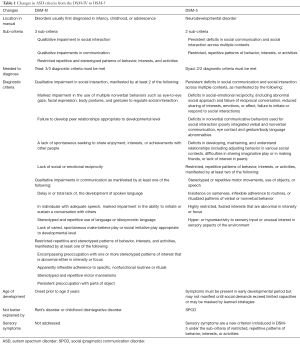
Full table
In DSM-5, the concept of a “spectrum” ASD diagnosis was created, combining the DSM-IV’s separate pervasive developmental disorder (PDD) diagnoses: autistic disorder, Asperger’s disorder, childhood disintegrative disorder, and pervasive developmental disorder not otherwise specified (PDD-NOS), into one. Rett syndrome is no longer included under ASD in DSM-5 as it is considered a discrete neurological disorder. A separate social (pragmatic) communication disorder (SPCD) was established for those with disabilities in social communication, but lacking repetitive, restricted behaviors. Additionally, severity level descriptors were added to help categorize the level of support needed by an individual with ASD.
This new definition is intended to be more accurate and works toward diagnosing ASD at an earlier age (3). However, studies estimating the potential impact of moving from the DSM-IV to the DSM-5 have predicted a decrease in ASD prevalence (4,5) and there has been concern that children with a previous PDD-NOS diagnosis would not meet criteria for ASD diagnosis (5-7). There are varying reports estimating the extent of and effects of this change. One study found that with parental report of ASD symptoms alone, the DSM-5 criteria identified 91% of children with clinical DSM-IV PDD diagnoses (8). However, a systematic review suggests only 50% to 75% of individuals maintain diagnoses (9) and other studies have also suggested a decreased rate of diagnosis of individuals with ASD under the DSM-5 criteria (10). Often those who did not meet the requirements were previously classified as high functioning Asperger’s syndrome and PDD-NOS (11,12). Overall, most studies suggest that the DSM-5 provides increased specificity and decreased sensitivity compared to the DSM-IV (5,13); so while those diagnosed with ASD are more likely to have the condition, there is a higher number of children whose ASD diagnosis is missed, particularly older children, adolescents, adults, or those with a former diagnosis of Asperger’s disorder or PDD-NOS (14). Nevertheless, the number of people who would be diagnosed under the DSM-IV, but not under the new DSM-5 appears to be declining over time, likely due to increased awareness and better documentation of behaviors (4).
It has yet to be determined how the new diagnosis of SPCD will impact the prevalence of ASD. One study found the new SPCD diagnosis encompasses those individuals who possess subthreshold autistic traits and do not qualify for a diagnosis of ASD, but who still have substantial needs (15). Furthermore, children who previously met criteria for PDD-NOS under the DSM-IV might now be diagnosed with SPCD.
Epidemiology
The World Health Organization (WHO) estimates the international prevalence of ASD at 0.76%; however, this only accounts for approximately 16% of the global child population (16). The Centers for Disease Control and Prevention (CDC) estimates about 1.68% of United States (US) children aged 8 years (or 1 in 59 children) are diagnosed with ASD (6,17). In the US, parent-reported ASD diagnoses in 2016 averaged slightly higher at 2.5% (18). The prevalence of ASD in the US more than doubled between 2000–2002 and 2010–2012 according to Autism and Developmental Disabilities Monitoring Network (ADDM) estimates (6). Although it may be too early to comment on trends, in the US, the prevalence of ASD has appeared to stabilize with no statistically significant increase from 2014 to 2016 (19). Changing diagnostic criteria may impact prevalence and the full impact of the DSM-5 diagnostic criteria has yet to be seen (17).
Insurance mandates requiring commercial plans to cover services for ASD along with improved awareness have likely contributed to the increase in ASD prevalence estimates as well as the increased diagnosis of milder cases of ASD in the US (6,20,21). While there was only a modest increase in prevalence immediately after the mandates, there have been additional increases later as health care professionals better understood the regulatory and reimbursement process. The increase in prevalence may also be due to changes in reporting practices. One study in Denmark found the majority of increase in ASD prevalence from 1980–1991 was based on changes of diagnostic criteria and inclusion of outpatient data, rather than a true increase in ASD prevalence (21).
ASD occurs in all racial, ethnic, and socioeconomic groups, but its diagnosis is far from uniform across these groups. Caucasian children are consistently identified with ASD more often than black or Hispanic children (6). While the differences appear to be decreasing, the continued discrepancy may be due to stigma, lack of access to healthcare services, and a patient’s primary language being one other than English.
ASD is more common in males (22,23) but in a recent meta-analysis (24), true male-to-female ratio is closer to 3:1 than the previously reported 4:1, though this study was not done using the DSM-5 criteria. This study also suggested that girls who meet criteria for ASD are at higher risk of not receiving a clinical diagnosis. The female autism phenotype may play a role in girls being misdiagnosed, diagnosed later, or overlooked. Not only are females less likely to present with overt symptoms, they are more likely to mask their social deficits through a process called “camouflaging”, further hindering a timely diagnosis (25). Likewise, gender biases and stereotypes of ASD as a male disorder could also hamper diagnoses in girls (26).
Several genetic diagnoses have an increased rate of co-occurring ASD compared to the average population, including fragile X, tuberous sclerosis, Down syndrome, Rett syndrome, among others; however, these known genetic disorders account for a very small amount of overall ASD cases (27-30). Studies of children with sex chromosome aneuploidy describe a specific social functioning profile in males that suggests more vulnerability to autism (22,23,31,32). With the increased use of chromosomal microarray, several sites (chromosome X, 2, 3, 7, 15, 16, 17, and 22 in particular) have proven to be associated with increased ASD risk (28).
Other risk factors for ASD include increased parental age and prematurity (33-35). This could be due to the theory that older gametes have a higher probability of carrying mutations which could result in additional obstetrical complications, including prematurity (36).
Causes
ASD is a neurobiological disorder influenced by both genetic and environmental factors affecting the developing brain. Ongoing research continues to deepen our understanding of potential etiologic mechanisms in ASD, but currently no single unifying cause has been elucidated.
Neuropathologic studies are limited, but have revealed differences in cerebellar architecture and connectivity, limbic system abnormalities, and frontal and temporal lobe cortical alterations, along with other subtle malformations (28,37,38). A small explorative study of neocortical architecture from young children revealed focal disruption of cortical laminar architecture in the majority of subjects, suggesting problems with cortical layer formation and neuronal differentiation (39). Brain overgrowth both in terms of cortical size and additionally in terms of increased extra-axial fluid have been described in children with ASD and are areas of ongoing study both in terms of furthering our understanding of its etiology, but also as a potential biomarker (40,41).
Genetic factors play a role in ASD susceptibility, with siblings of patients with ASD carrying an increased risk of diagnosis when compared to population norms, and a much higher, although not absolute, concordance of autism diagnosis in monozygotic twins (42-44).
Genome wide association studies and whole exome sequencing methods have broadened our understanding of ASD susceptibility genes, and learning more regarding the function of these genes can shed light on potential biologic mechanisms (45). For example candidate genes in ASD include those that play a role in brain development or neurotransmitter function, or genes that affect neuronal excitability (46,47). Many of the genetic defects associated with ASD encode proteins that are relevant at the neuronal synapse or that are involved in activity-dependent changes in neurons, including regulatory proteins such as transcription factors (42,48). Potential “networks” of ASD genetic risk convergence include pathways involved in neurotransmission and neuroinflammation (49). Transcriptional and splicing dysregulation or alterations in epigenetic mechanisms such as DNA methylation or histone acetylation and modification may play a role (42,49-51). A recent study describes 16 newly identified genes associated with ASD that raise new potential mechanisms including cellular cytoskeletal structure and ion transport (52). Ultimately, ASD remains one of the most genetically heterogeneous neuropsychiatric disorders with rarer de novo and inherited variants in over 700 genes (53).
While genetics clearly play a role in ASD’s etiology, phenotypic expression of genetic susceptibility remains extremely variable within ASD (54). Genetic risk may be modulated by prenatal, perinatal, and postnatal environmental factors in some patients (35). Prenatal exposure to thalidomide and valproic acid have been reported to increase risk, while studies suggest that prenatal supplements of folic acid in patients exposed to antiepileptic drugs may reduce risk (55-57). Research has not confirmed if a small positive trial of folinic acid in autism can be used to recommend supplementation more broadly (58). Advanced maternal and paternal age have both been shown to have an increased risk of having a child with ASD (59). Maternal history of autoimmune disease, such as diabetes, thyroid disease, or psoriasis has been postulated, but study results remain mixed (60,61). Maternal infection or immune activation during pregnancy is another area of interest and may be a potential risk factor according to recent investigations (62-65). Both shorter and longer inter-pregnancy intervals have also been reported to increase ASD risk (66). Infants born prematurely have been demonstrated to carry a higher risk for ASD in addition to other neurodevelopmental disorders (34). In a prior epidemiologic review, obstetric factors including uterine bleeding, caesarian delivery, low birthweight, preterm delivery, and low Apgar scores were reported to be the few factors more consistently associated with autism (67). A recent meta-analysis reported several pre, peri and postnatal risk factors that resulted in an elevated relative risk of ASD in offspring (35), but also revealed significant heterogeneity, resulting in an inability to make true determination regarding the importance of these factors.
Despite the hysteria surrounding the now retracted Lancet article first published in 1998, there is no evidence that vaccines, thimerosal, or mercury is associated with ASD (68-70). In the largest single study to date, there was not an increased risk after measles/mumps/rubella (MMR) vaccination in a nationwide cohort study of Danish children (70).
Ultimately, research continues to reveal factors that correlate with ASD risk, but no causal determinations have been made. This leaves much room for discovery with investigators continuing to elucidate new variants conveying genetic risk, or new environmental correlates that require further study (52).
Evaluation
Evaluation in ASD begins with screening of the general pediatric population to identify children at-risk or demonstrating signs suggestive of ASD, following which a diagnostic evaluation is recommended. The American Academy of Pediatrics (AAP) guidelines recommend developmental surveillance at 9, 15 and 30 months well child visits and autism specific screening at 18 months and again at 24 or 30 months (28,71). Early red flags for ASD include poor eye contact, poor response to name, lack of showing and sharing, no gesturing by 12 months, and loss of language or social skills. Screening tools for ASD in this population include the Modified Checklist for Autism in Toddlers, Revised, with Follow-up (M-CHAT-R/F) and Survey of Wellbeing of Young Children (SWYC) (72,73). Red flags in preschoolers may include limited pretend play, odd or intensely focused interests, and rigidity. School age children may demonstrate concrete or literal thinking, have trouble understanding emotions, and may even show an interest in peers but lack conversational skills or appropriate social approach. If there is suspicion of ASD in these groups, screening tools available include the Social Communication Questionnaire (SCQ), Social Responsiveness Scale (SRS), and Autism Spectrum Screening Questionnaire (ASSQ) (74-76).
If concerns are raised at screening, primary care clinicians are recommended to refer the child to early intervention if less than 3 years of age or to the public school system for psychoeducational evaluation in order to establish an individual education program (IEP) if the child is three years of age or older. Clinicians should additionally refer the child to a specialist (pediatric neurologist, developmental-behavioral pediatrician, child psychiatrist, licensed child psychologist) for a definitive diagnosis and comprehensive assessment (71). A comprehensive assessment should include a complete physical exam, including assessment for dysmorphic features, a full neurologic examination with head circumference, and a Wood’s lamp examination of the skin. A parent interview, collection of any outside informant observations, and a direct clinician observation of the child’s current cognitive, language, and adaptive functioning by a clinician experienced with ASD should be components of this comprehensive assessment. (28,71,77,78).
Additionally, primary care clinicians need to be aware of (and evaluate for) potential co-occurring conditions in children with ASD. According to a surveillance study of over 2,000 children with ASD, 83% had an additional developmental diagnosis, 10% had at least one psychiatric diagnosis, and 16% at least one neurologic diagnosis (79). In the past, rates of co-morbid intellectual disability (ID) in patients with ASD were reported from 50% to 70%, with the most recent CDC estimate reported at 31.0% (26.7% to 39.4%) with ID defined as intelligence quotient (IQ) ≤70 (6,80). Other common co-occurring medical conditions include gastrointestinal (GI) disorders, including dietary restrictions and food selectivity, sleep disorders, obesity, and seizures (81-84). Studies using electronic health record (EHR) analysis revealed prevalence of epilepsy ~20% and GI disorders [without inflammatory bowel disease (IBD)] at 10–12% (82). Epilepsy has been shown to have higher prevalence rates in ASD with comorbid ID and medical disorders of increased risk such as tuberous sclerosis complex (TSC) (85-87). GI disorders or GI symptomatology, including diarrhea, constipation, restrictive eating, or reflux, have been shown to be prominent in ASD across multiple studies (81,82,88,89). Sleep problems have been reported to occur in anywhere from 50% to 73% of patients with ASD with variation in prevalence dependent on the definition of sleep symptoms or the measurement tool used (90-92). Rates of overweight and obesity in ASD are reported to be roughly 33% and 18% respectively, higher than rates in typically developing children (81-84,93).
Other behavioral or psychiatric co-occurring conditions in ASD include anxiety, attention deficit/hyperactivity disorder (ADHD), obsessive compulsive disorder, and mood disorders or other disruptive behavior disorders (81). Rates of co-occurring ADHD are reported anywhere from 25% to 81% (81,94). A recent meta-analysis of 30 studies measuring rates of anxiety and 29 studies measuring rates of depression reported a high degree of heterogeneity from the current literature, but stated pooled lifetime prevalence for adults with ASD to be 42% for any anxiety disorder and 37% for any depressive disorder, though the use of self-report measures and the presence of ID could influence estimates (95). In children with ASD seeking treatment, the rate of any anxiety disorder was found to be similar at 42% and in addition this study reported co-morbid oppositional defiant disorder at a rate of 46% and mood disorders at 8%, with 66% of the sample of over 600 patients having more than one co-occurring condition (94).
Currently no clear ASD biomarkers or diagnostic measures exist, and the diagnosis is made based on fulfillment of descriptive criteria. In light of a relatively high yield in patients with ASD, clinical genetic testing is recommended and can provide information regarding medical interventions or work up that might be necessary and help with family planning (96). The American College of Medical Genetics and Genomics (ACMGG) guidelines currently recommend chromosomal microarray for all children, fragile X testing in males, and additional gene sequencing, including PTEN and MECP2, in certain patients as first tier genetic testing in the work up of ASD (97). High resolution G-banded karyotype, once recommended for all patients with ASD, is no longer routinely indicated based on recent consensus recommendations, but might still be performed in patients with a family or reproductive history suggestive of chromosomal rearrangements or specific syndromes such as sex chromosome anomalies or Trisomy 21 (96-98). Several professional societies recommend genetic testing for ASD, including the American Academy of Neurology, the AAP, ACMGG, and the American Academy of Child and Adolescent Psychiatry, and a child may require further referral to a geneticist and/or genetic counselor, depending on results of testing (25,28,97,99). As the field of genetics continues to advance rapidly, recent publications suggest whole exome sequencing may become the preferred method for clinical genetic testing in individuals with ASD (100,101).
Aside from genetic testing, no other laboratory work up is routinely recommended for every patient with a diagnosis of ASD. However, further evaluation may be appropriate for patients with particular findings or risk factors. Metabolic work-up should be considered in patients with any of the following concerning symptoms or signs: a history of clear developmental regression including loss or plateau of motor skills; hypotonia; recurrent episodes of vomiting, lethargy or hypoglycemia; microcephaly or poor growth; concern for other organ involvement; coarse features; or concern for seizures or ataxia. Based on the patient’s history and presentation, components of a metabolic laboratory evaluation could include complete blood count (CBC), liver and renal function tests, lactate, pyruvate, carnitine, amino acids, an acylcarnitine profile, urine organic acids and/or urine glycosaminoglycans (97,102). Children with a history of pica should have a lead level measured (28,103). In a child with significantly restricted food intake, one should consider a laboratory evaluation of nutritional status. Sleep symptoms may warrant a referral for a possible sleep study, and if restless sleep symptoms are present, an evaluation for iron deficiency is not unreasonable, particularly if dietary rigidity limits iron intake (104).
Neuroimaging is not routinely recommended for every patient with ASD (28,99), but may be appropriate in patients with a suspicion for TSC or other neurocutaneous disorders, microcephaly, or an abnormal neurologic exam (spasticity, severe hypotonia, unilateral findings). Patients with suspected seizures should have an electroencephalography (EEG) obtained (102). If accessible, it might be appropriate to immediately refer children with concern for further genetic, metabolic or neurologic conditions to a specialist who can then obtain and interpret the aforementioned testing. At this time there is inadequate evidence to recommend routine testing for celiac disease, immunologic or neurochemical markers, mitochondrial disorders, allergy testing, hair analysis, intestinal permeability studies, erythrocyte glutathione peroxidase studies, stool analysis, urinary peptides or vitamin and mineral deficiencies without a history of severe food selectivity.
Summary
ASD is a neurodevelopmental disorder characterized by deficits in social communication and the presence of restricted interests and repetitive behaviors. Recent changes to the diagnostic criteria occurred with the transition to the new diagnostic manual (DSM-5) and will likely impact prevalence, which currently stands at 1 in 59 children in the US. ASD is a neurobiological disorder influenced by both genetic and environmental factors affecting the developing brain. Research continues to reveal factors that correlate with ASD risk and these findings may guide further etiologic investigation, but no final causal pathway has been elucidated. Clinical evaluation begins with developmental screening of the general pediatric population to identify at-risk children, followed by referral to a specialist for a definitive diagnosis and comprehensive neuropsychological assessment. Children with ASD should also be screened for common co-morbid diagnoses. While no clear biomarkers or diagnostic measures exist, clinical genetic testing is recommended as part of the initial medical evaluation. Further medical work up or subspecialist referrals may be pursued based on specific patient characteristics.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5th ed. Arlington: American Psychiatric Publishing, 2013.
- American Psychiatric Association. Diagnostic and statistcal manual of mental disorders. 4th ed. Washington: American Psychiatric Publishing, 1994.
- Halfon N, Kuo AA. What DSM-5 could mean to children with autism and their families. JAMA Pediatr 2013;167:608-13. [Crossref] [PubMed]
- Maenner MJ, Rice CE, Arneson CL, et al. Potential impact of DSM-5 criteria on autism spectrum disorder prevalence estimates. JAMA Psychiatry 2014;71:292-300. [Crossref] [PubMed]
- Kulage KM, Smaldone AM, Cohn EG. How will DSM-5 affect autism diagnosis? A systematic literature review and meta-analysis. J Autism Dev Disord 2014;44:1918-32. [Crossref] [PubMed]
- Baio J, Wiggins L, Christensen DL, et al. Prevalence of autism spectrum disorder among children aged 8 years — autism and developmental disabilities monitoring network, 11 sites, United States, 2014. MMWR Surveill Summ 2018;67:1-23. [Crossref] [PubMed]
- Yaylaci F, Miral S. A comparison of DSM-IV-TR and DSM-5 diagnostic classifications in the clinical diagnosis of autistic spectrum disorder. J Autism Dev Disord 2017;47:101-9. [Crossref] [PubMed]
- Huerta M, Bishop SL, Duncan A, et al. Application of DSM-5 criteria for autism spectrum disorder to three samples of children with DSM-IV diagnoses of pervasive developmental disorders. Am J Psychiatry 2012;169:1056-64. [Crossref] [PubMed]
- Sturmey P, Dalfern S. The effects of DSM5 autism diagnostic criteria on number of individuals diagnosed with autism spectrum disorders: a systematic review. Rev J Autism Dev Disord 2014;1:249-52. [Crossref]
- Hartley-McAndrew M, Mertz J, Hoffman M, et al. Rates of autism spectrum disorder diagnosis under the DSM-5 criteria compared to DSM-IV-TR criteria in a hospital-based clinic. Pediatr Neurol 2016;57:34-8. [Crossref] [PubMed]
- de Giambattista C, Ventura P, Trerotoli P, et al. Subtyping the autism spectrum disorder: comparison of children with high functioning autism and asperger syndrome. J Autism Dev Disord 2019;49:138-50. [Crossref] [PubMed]
- Mazurek MO, Lu F, Symecko H, et al. A prospective study of the concordance of DSM-IV and DSM-5 diagnostic criteria for autism spectrum disorder. J Autism Dev Disord 2017;47:2783-94. [Crossref] [PubMed]
- Gibbs V, Aldridge F, Chandler F, et al. Brief report: an exploratory study comparing diagnostic outcomes for autism spectrum disorders under DSM-IV-TR with the proposed DSM-5 revision. J Autism Dev Disord 2012;42:1750-6. [Crossref] [PubMed]
- Lai MC, Lombardo MV, Chakrabarti B, et al. Subgrouping the autism "spectrum": reflections on DSM-5. PLoS Biol 2013;11:e1001544. [Crossref] [PubMed]
- Mandy W, Wang A, Lee I, et al. Evaluating social (pragmatic) communication disorder. J Child Psychol Psychiatry 2017;58:1166-75. [Crossref] [PubMed]
- Baxter AJ, Brugha TS, Erskine HE, et al. The epidemiology and global burden of autism spectrum disorders. Psychol Med 2015;45:601-13. [Crossref] [PubMed]
- Palinkas LA, Mendon SJ, Hamilton AB. Annual review of public health innovations in mixed methods evaluations. Annu Rev Public Heal 2019;40:423-42. [Crossref]
- Kogan MD, Vladutiu CJ, Schieve LA, et al. The prevalence of parent-reported autism spectrum disorder among US children. Pediatrics 2018;142:e20174161. [Crossref] [PubMed]
- Xu G, Strathearn L, Liu B, et al. Prevalence of autism spectrum disorder among US children and adolescents, 2014-2016. JAMA 2018;319:81. [Crossref] [PubMed]
- Mandell DS, Barry CL, Marcus SC, et al. Effects of autism spectrum disorder insurance mandates on the treated prevalence of autism spectrum disorder. JAMA Pediatr 2016;170:887-93. [Crossref] [PubMed]
- Hansen SN, Schendel DE, Parner ET. Explaining the increase in the prevalence of autism spectrum disorders: the proportion attributable to changes in reporting practices. JAMA Pediatr 2015;169:56-62. [Crossref] [PubMed]
- Demily C, Poisson A, Peyroux E, et al. Autism spectrum disorder associated with 49,XYYYY: case report and review of the literature. BMC Med Genet 2017;18:9. [Crossref] [PubMed]
- Tartaglia NR, Wilson R, Miller JS, et al. Autism spectrum disorder in males with sex chromosome aneuploidy: XXY/klinefelter syndrome, XYY, and XXYY. J Dev Behav Pediatr 2017;38:197-207. [Crossref] [PubMed]
- Loomes R, Hull L, Mandy WPL. What is the male-to-female ratio in autism spectrum disorder? A systematic review and meta-analysis. J Am Acad Child Adolesc Psychiatry 2017;56:466-74. [Crossref] [PubMed]
- Volkmar F, Siegel M, Woodbury-Smith M, et al. Practice parameter for the assessment and treatment of children and adolescents with autism spectrum disorder. J Am Acad Child Adolesc Psychiatry 2014;53:237-57. [Crossref] [PubMed]
- Bargiela S, Steward R, Mandy W. The experiences of late-diagnosed women with autism spectrum conditions: an investigation of the female autism phenotype. J Autism Dev Disord 2016;46:3281-94. [Crossref] [PubMed]
- Sztainberg Y, Zoghbi HY. Lessons learned from studying syndromic autism spectrum disorders. Nat Neurosci 2016;19:1408-17. [Crossref] [PubMed]
- Johnson CP, Myers SM. Identification and evaluation of children with autism spectrum disorders. Pediatrics 2007;120:1183-215. [Crossref] [PubMed]
- Reddy KS. Cytogenetic abnormalities and fragile-X syndrome in autism spectrum disorder. BMC Med Genet 2005;6:3. [Crossref] [PubMed]
- Yoo H. Genetics of autism spectrum disorder: current status and possible clinical applications. Exp Neurobiol 2015;24:257. [Crossref] [PubMed]
- Ross JL, Roeltgen DP, Kushner H, et al. Behavioral and social phenotypes in boys with 47,XYY syndrome or 47,XXY klinefelter syndrome. Pediatrics 2012;129:769-78. [Crossref] [PubMed]
- Bardsley MZ, Kowal K, Levy C, et al. 47,XYY syndrome: clinical phenotype and timing of ascertainment. J Pediatr 2013;163:1085-94. [Crossref] [PubMed]
- Durkin MS, Maenner MJ, Newschaffer CJ, et al. Advanced parental age and the risk of autism spectrum disorder. Am J Epidemiol 2008;168:1268-76. [Crossref] [PubMed]
- Agrawal S, Rao SC, Bulsara MK, et al. Prevalence of autism spectrum disorder in preterm infants: a meta-analysis. Pediatrics 2018;142:e20180134. [Crossref] [PubMed]
- Wang C, Geng H, Liu W, et al. Prenatal, perinatal, and postnatal factors associated with autism: a meta-analysis. Medicine (Baltimore) 2017;96:e6696. [Crossref] [PubMed]
- Parner ET, Baron-Cohen S, Lauritsen MB, et al. Parental age and autism spectrum disorders. Ann Epidemiol 2012;22:143-50. [Crossref] [PubMed]
- Skefos J, Cummings C, Enzer K, et al. Regional alterations in Purkinje cell density in patients with autism. PLoS One 2014;9:e81255. [Crossref] [PubMed]
- Stoodley CJ, D’Mello AM, Ellegood J, et al. Altered cerebellar connectivity in autism and cerebellar-mediated rescue of autism-related behaviors in mice. Nat Neurosci 2017;20:1744-51. [Crossref] [PubMed]
- De Rubeis S, He X, Goldberg AP, et al. Synaptic, transcriptional and chromatin genes disrupted in autism. Nature 2014;515:209-15. [Crossref] [PubMed]
- Shen MD, Kim SH, McKinstry RC, et al. Increased extra-axial cerebrospinal fluid in high-risk infants who later develop autism. Biol Psychiatry 2017;82:186-93. [Crossref] [PubMed]
- Hazlett HC, Gu H, Munsell BC, et al. Early brain development in infants at high risk for autism spectrum disorder. Nature 2017;542:348-51. [Crossref] [PubMed]
- Kim H, Keifer C, Rodriguez-Seijas C, et al. Quantifying the optimal structure of the autism phenotype: a comprehensive comparison of dimensional, categorical, and hybrid models. J Am Acad Child Adolesc Psychiatry 2019;58:876-86.e2. [Crossref] [PubMed]
- Sandin S, Lichtenstein P, Kuja-Halkola R, et al. The familial risk of autism. JAMA 2014;311:1770-7. [Crossref] [PubMed]
- Risch N, Hoffmann TJ, Anderson M, et al. Familial recurrence of autism spectrum disorder: Evaluating genetic and environmental contributions. Am J Psychiatry 2014;171:1206-13. [Crossref] [PubMed]
- Walsh CA, Morrow EM, Rubenstein JLR. Autism and brain development. Cell 2008;135:396-400. [Crossref] [PubMed]
- Rubenstein JL, Merzenich MM. Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes Brain Behav 2003;2:255-67. [Crossref] [PubMed]
- McDougle CJ, Erickson CA, Stigler KA, et al. Neurochemistry in the pathophysiology of autism. J Clin Psychiatry 2005;66 Suppl 10:9-18. [PubMed]
- Zoghbi HY. Postnatal neurodevelopmental disorders: meeting at the synapse? Science 2003;302:826-30. [Crossref] [PubMed]
- Voineagu I, Wang X, Johnston P, et al. Transcriptomic analysis of autistic brain reveals convergent molecular pathology. Nature 2011;474:380-4. [Crossref] [PubMed]
- Ladd-Acosta C, Hansen KD, Briem E, et al. Common DNA methylation alterations in multiple brain regions in autism. Mol Psychiatry 2014;19:862-71. [Crossref] [PubMed]
- Sun W, Poschmann J, Cruz-Herrera Del Rosario R, et al. Histone acetylome-wide association study of autism spectrum disorder. Cell 2016;167:1385-97.e11. [Crossref] [PubMed]
- Ruzzo EK, Pérez-Cano L, Jung JY, et al. Inherited and de novo genetic risk for autism impacts shared networks. Cell 2019;178:850-66.e26. [Crossref] [PubMed]
- Saxena A, Chahrour M. Autism spectrum disorder. In: Ginsburg GS, Willard HF, David SP. editors. Genomic and precision medicine: primary care. Cambridge: Academic Press, 2017:301-16.
- Veenstra-Vanderweele J, Christian SL, Cook EH Jr. Autism as a paradigmatic complex genetic disorder. Annu Rev Genomics Hum Genet 2004;5:379-405. [Crossref] [PubMed]
- Surén P, Roth C, Bresnahan M, et al. Association between maternal use of folic acid supplements and risk of autism spectrum disorders in children. JAMA 2013;309:570-7. [Crossref] [PubMed]
- Bjørk M, Riedel B, Spigset O, et al. Association of folic acid supplementation during pregnancy with the risk of autistic traits in children exposed to antiepileptic drugs in utero. JAMA Neurol 2018;75:160. [Crossref] [PubMed]
- Rasalam AD, Hailey H, Williams JHG, et al. Characteristics of fetal anticonvulsant syndrome associated autistic disorder. Dev Med Child Neurol 2005;47:551-5. [Crossref] [PubMed]
- Frye RE, Slattery J, Delhey L, et al. Folinic acid improves verbal communication in children with autism and language impairment: a randomized double-blind placebo-controlled trial. Mol Psychiatry 2018;23:247-56. [Crossref] [PubMed]
- Croen LA, Najjar DV, Fireman B, et al. Maternal and paternal age and risk of autism spectrum disorders. Arch Pediatr Adolesc Med 2007;161:334-40. [Crossref] [PubMed]
- Xiang AH, Wang X, Martinez MP, et al. Maternal type 1 diabetes and risk of autism in offspring. JAMA 2018;320:89-91. [Crossref] [PubMed]
- Croen LA, Grether JK, Yoshida CK, et al. Maternal autoimmune diseases, asthma and allergies, and childhood autism spectrum disorders: a case-control study. Arch Pediatr Adolesc Med 2005;159:151-7. [Crossref] [PubMed]
- Malkova NV, Yu CZ, Hsiao EY, et al. Maternal immune activation yields offspring displaying mouse versions of the three core symptoms of autism. Brain Behav Immun 2012;26:607-16. [Crossref] [PubMed]
- Estes ML, McAllister AK. Immune mediators in the brain and peripheral tissues in autism spectrum disorder. Nat Rev Neurosci 2015;16:469-86. [Crossref] [PubMed]
- Choi GB, Yim YS, Wong H, et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 2016;351:933-9. [Crossref] [PubMed]
- Croen LA, Qian Y, Ashwood P, et al. Infection and fever in pregnancy and autism spectrum disorders: findings from the study to explore early development. Autism Res 2019. [Epub ahead of print]. [PubMed]
- Schieve LA, Tian LH, Drews-Botsch C, et al. Autism spectrum disorder and birth spacing: findings from the study to explore early development (SEED). Autism Res 2018;11:81-94. [Crossref] [PubMed]
- Newschaffer CJ, Croen LA, Daniels J, et al. The epidemiology of autism spectrum disorders. Annu Rev Public Health 2007;28:235-58. [Crossref] [PubMed]
- Taylor LE, Swerdfeger AL, Eslick GD. Vaccines are not associated with autism: an evidence-based meta-analysis of case-control and cohort studies. Vaccine 2014;32:3623-9. [Crossref] [PubMed]
- Uno Y, Uchiyama T, Kurosawa M, et al. Early exposure to the combined measles-mumps-rubella vaccine and thimerosal-containing vaccines and risk of autism spectrum disorder. Vaccine 2015;33:2511-6. [Crossref] [PubMed]
- Hviid A, Hansen JV, Frisch M, et al. Measles, mumps, rubella vaccination and autism a nationwide cohort study. Ann Intern Med 2019;170:513-20. [Crossref] [PubMed]
- Ellerbeck K, Smith C, Courtemanche A. Care of children with autism spectrum disorder. Prim Care 2015;42:85-98. [Crossref] [PubMed]
- Robins DL, Casagrande K, Barton M, et al. Validation of the modified checklist for autism in toddlers, revised with follow-up (M-CHAT-R/F). Pediatrics 2014;133:37-45. [Crossref] [PubMed]
- Smith NJ, Sheldrick RC, Perrin EC. An abbreviated screening instrument for autism spectrum disorders. Infant Ment Health J 2013;34:149-55. [Crossref]
- Ehlers S, Gillberg C, Wing L. A screening questionnaire for Asperger syndrome and other high- functioning autism spectrum disorders in school age children. J Autism Dev Disord 1999;29:129-41. [Crossref] [PubMed]
- Rutter M, Bailey A, Lord CM. The social communication questionnaire. Los Angeles: Western Psychological Services, 2003.
- Constantino JN, Gruber CP. Social responsiveness scale (SRS). Los Angeles: Western Psychological Services, 2005.
- Huerta M, Lord C. Diagnostic evaluation of autism spectrum disorders. Pediatr Clin North Am 2012;59:103-11. [Crossref] [PubMed]
- Lord C, Rutter M, DiLavore P, et al. Autism diagnostic observation schedule, second edition. Torrance: Western Psychological Services, 2012.
- Levy SE, Giarelli E, Lee LC, et al. Autism spectrum disorder and co-occurring developmental, psychiatric, and medical conditions among children in multiple populations of the United States. J Dev Behav Pediatr 2010;31:267-75. [Crossref] [PubMed]
- Yeargin-Allsopp M, Rice C, Karapurkar T, et al. Prevalence of autism in a US metropolitan area. JAMA 2003;289:49-55. [Crossref] [PubMed]
- Soke GN, Maenner MJ, Christensen D, et al. Prevalence of co-occurring medical and behavioral conditions/symptoms among 4- and 8-year-old children with autism spectrum disorder in selected areas of the United States in 2010. J Autism Dev Disord 2018;48:2663-76. [Crossref] [PubMed]
- Kohane IS, McMurry A, Weber G, et al. The co-morbidity burden of children and young adults with autism spectrum disorders. PLoS One 2012;7:e33224. [Crossref] [PubMed]
- Kielinen M, Rantala H, Timonen E, et al. Associated medical disorders and disabilities in children with autistic disorder: A population-based study. Autism 2004;8:49-60. [Crossref] [PubMed]
- Curtin C, Anderson SE, Must A, et al. The prevalence of obesity in children with autism: a secondary data analysis using nationally representative data from the National Survey of Children’s Health. BMC Pediatr 2010;10:11. [Crossref] [PubMed]
- Tuchman R, Rapin I. Epilepsy in autism. Lancet Neurol 2002;1:352-8. [Crossref] [PubMed]
- Ewen JB, Marvin AR, Law K, et al. Epilepsy and autism severity: a study of 6,975 children. Autism Res 2019;12:1251-9. [Crossref] [PubMed]
- Amiet C, Gourfinkel-An I, Bouzamondo A, et al. Epilepsy in autism is associated with intellectual disability and gender: evidence from a meta-analysis. Biol Psychiatry 2008;64:577-82. [Crossref] [PubMed]
- Alabaf S, Gillberg C, Lundström S, et al. Physical health in children with neurodevelopmental disorders. J Autism Dev Disord 2019;49:83-95. [Crossref] [PubMed]
- Ibrahim SH, Voigt RG, Katusic SK, et al. Incidence of gastrointestinal symptoms in children with autism: a population-based study. Pediatrics 2009;124:680-6. [Crossref] [PubMed]
- Reynolds AM, Soke GN, Sabourin KR, et al. Sleep problems in 2- to 5-year-olds with autism spectrum disorder and other developmental delays. Pediatrics 2019;143:e20180492. [Crossref] [PubMed]
- Krakowiak P, Goodlin-Jones B, Hertz-Picciotto I, et al. Sleep problems in children with autism spectrum disorders, developmental delays, and typical development: a population-based study. J Sleep Res 2008;17:197-206. [Crossref] [PubMed]
- Polimeni MA, Richdale AL, Francis AJ. A survey of sleep problems in autism, Asperger's disorder and typically developing children. J Intellect Disabil Res 2005;49:260-8. [Crossref] [PubMed]
- Hill AP, Zuckerman KE, Fombonne E. Obesity and autism. Pediatrics 2015;136:1051-61. [Crossref] [PubMed]
- Lecavalier L, McCracken CE, Aman MG, et al. An exploration of concomitant psychiatric disorders in children with autism spectrum disorder. Compr Psychiatry 2019;88:57-64. [Crossref] [PubMed]
- Hollocks MJ, Lerh JW, Magiati I, et al. Anxiety and depression in adults with autism spectrum disorder: a systematic review and meta-analysis. Psychol Med 2019;49:559-72. [Crossref] [PubMed]
- Miller DT. Genetic testing for autism: recent advances and clinical implications. Expert Rev Mol Diagn 2010;10:837-40. [Crossref] [PubMed]
- Schaefer GB, Mendelsohn NJ. Clinical genetics evaluation in identifying the etiology of autism spectrum disorders: 2013 guideline revisions. Genet Med 2013;15:399-407. [Crossref] [PubMed]
- Shen Y, Dies KA, Holm IA, et al. Clinical genetic testing for patients with autism spectrum disorders. Pediatrics 2010;125:e727-35. [Crossref] [PubMed]
- Filipek PA, Accardo PJ, Ashwal S, et al. Practice parameter: screening and diagnosis of autism. Report of the quality standards subcommittee of the American Academy of Neurology and the Child Neurology Society. Neurology 2000;55:468-79. [Crossref] [PubMed]
- Srivastava S, Love-Nichols JA, Dies KA, et al. Meta-analysis and multidisciplinary consensus statement: exome sequencing is a first-tier clinical diagnostic test for individuals with neurodevelopmental disorders. Genet Med 2019. [Epub ahead of print]. [Crossref] [PubMed]
- Feliciano P, Zhou X, Astrovskaya I, et al. Exome sequencing of 457 autism families recruited online provides evidence for autism risk genes. NPJ Genom Med 2019;4:19. [Crossref] [PubMed]
- American Academy of Pediatrics. Autism – caring for children with autism spectrum disorders: a resource toolkit for clinicians. 3rd ed. Itasca: American Academy of Pediatrics, 2019.
- Shannon M, Graef JW. Lead intoxication in children with pervasive developmental disorders. J Toxicol Clin Toxicol 1996;34:177-81. [Crossref] [PubMed]
- Dosman C, Witmans M, Zwaigenbaum L. Iron's role in paediatric restless legs syndrome - a review. Paediatr Child Health 2012;17:193-7. [Crossref] [PubMed]


