
Early evaluation of the metabolic syndrome in Bucaramanga, Colombia
Introduction
Metabolic syndrome (MS) was initially described as a combination of cardiovascular risk factors such as high triglycerides levels, low HDL cholesterol levels, high blood pressure and abdominal obesity associated to insulin resistance, whose grouped components were associated to an increase in cardiometabolic risk (1). Unlike in the pediatric population, there are well defined diagnostic criteria in the adult population (2). Only until 2007 the International Diabetes Federation (IDF) through a consensus of experts, considered abdominal obesity as the main component of the MS along with other two risk factors. This consensus only considers children between the ages of 10 and 15 (3) without the existence of clear criteria applicable to other age groups. MS diagnosis in children and adults tends to be done with the same criterion which consists on meeting at least three out of the aforementioned risk factors, although with some variations in the cut points or the presence of abdominal obesity along with other two risk factors (4,5).
The MS is considered a major public health issue worldwide since people with this disease have greater risk of developing type 2 diabetes, cardiovascular diseases (CVD), and kidney impairment, hence the interest in diagnosing and intervening the childhood population in an early manner (4,6). Furthermore, presence of MS during puberty in girls affects their fertility in adulthood (7,8). Likewise, presence of MS in boys has been associated with the diagnosis of obesity and its complications in the adult age (9).
On the other hand, adolescence is a stage of great interest for the formulation of public health policies since it is a term in which attitudes and practices are established, especially the adoption of risk or protective behaviors in their adult life (10). It has been shown that behaviors acquired during the adolescence tend to perpetuate in the further stages of the life cycle (11). Certain behaviors of adolescents have been related to an increase in mortality associated to chronic diseases (11). Some of these behaviors include low levels of physical activity, smoking, noxious consumption of alcohol, and food habits that favor the ingestion of ultra-processed foods with high contents of sugar, fat, and salt (12).
Thus, the increase in the prevalence of associated factors to the development of type 2 diabetes mellitus and CVD in children and adolescents in developing countries has awaken great interest to achieve a better understanding of their epidemiology and impact in clinical practice and public health. In Colombia, the largest population based cohort so far has characterized the school age population from the sociodemographic, clinical, metabolic, nutritional, and physical activity standpoints (13,14).
The presence of one or more criteria considered in the diagnosis of childhood MS in a developing country such as Colombia justifies the realization of follow-up studies on the midterm. The aim of this study was to compare the MS prevalence estimated in a representative sample of school age population in Bucaramanga, Colombia, and the MS prevalence estimated in a sub sample from the same population in the adolescent stage.
Methods
Design
Analytical cross-sectional study, nested in a population-based cohort.
Population and sample
The study population included a representative sample of boys and girls of school age residing in the city of Bucaramanga, Colombia from July 2006 to October 2007 (13). A sub sample of the same population was evaluated 10 years after (Barrera JG, 2012 unpublished data).
Data collection on the basal level and during follow-up
When establishing the cohort (population between 6 and 10 years of age) a house visit was conducted. During the visit, information related to sociodemographic, nutritional, and physical activities variables was collected. A complete physical check-up was conducted in a hospital visit (including anthropometry and nutritional assessment) and blood samples were taken for measuring fasting glycemia, lipid profile, uric acid, and insulin (13). At the time of follow-up, the same participants in their adolescence years (population between 14 to 19 years of age) were invited by means of telephone call to a hospital visit where we collected the same information obtained previously as baseline. They were given a complete medical assessment (including anthropometry) and blood samples were taken for measuring fasting glycemia, lipid profile, uric acid, and insulin (13). A house visit was conducted in order to perform the complete clinical assessment when the contact by phone was successful but participants did not go to the health institution (n=43 participants).
Data collection was carried out by physicians and nurses previously trained by the study researchers, using standardized forms. Data quality was reviewed in order to ensure that data were complete. Double typing of data was done on Excel software (Microsoft Corp., Redmond, WA, USA), and databases were compared using Epi-Info 2000. All discrepancies found were corrected by using the original data capture forms.
Outcomes of interest from the study
The main outcome was to define the MS by the two criteria mostly used worldwide for its diagnosis. Firstly, there are the Adult Treatment Panel III criteria (ATP III) (15), according to which three out of five criteria must be met (any of them): (I) abdominal obesity with waist perimeter > percentile 90, body mass index (BMI) percentile ≥85; (II) blood pressure > percentile 90; (III) triglycerides >110 mg/dL or percentile >95; (IV) HDL cholesterol <40 mg/dL in both sexes or percentile <5; (V) basal glucose (BG) >100 mg/dL or 2 hours after overload >140 mg/dL. Secondly, we have the criteria of the IDF (3) that define MS always taking into account the abdominal obesity and two of the other four criteria assessed: (I) abdominal obesity with waist perimeter > percentile 90; (II) blood pressure >130/85 mmHg; (III) triglycerides >150 mg/dL; (IV) HDL cholesterol <40 mg/dL in both sexes; (V) FG >100 mg/dL or 2 hours after overload >140 mg/dL. Other variables evaluated in this study can be found in supplementary material.
Statistical analysis
A descriptive analysis was conducted, where categorical values were presented as proportional, and the continuous ones as means and standard deviation, or means and interquartile range, according to their distribution. The differences between the data collected on the base level and during follow-up were determined by using Student’s t-test if the continuous variables had normal distribution as well as the differences between the diagnosis criteria employed (ATP III and IDF). Otherwise, the Mann Whitney U test was used. A Chi-squared test or Fisher’s exact test were employed for the categorical variables. All of the data were analyzed in the statistical software Stata version 14.0 (Stata Corporation: College Station, TX, USA).
Ethical considerations
The protocols of the original cohort and the follow-up study were reviewed and approved by the Committee for Ethics in Research of Fundación Cardiovascular de Colombia in act No. 396 of April 9, 2016. Consent was obtained from the parents or legal guardians of the participants by written. Additionally, all participants of school age gave their verbal assent and they signed an agreement (assent), and for the follow-up those teenagers of legal age (18 years) gave their written consent.
Results
The analytical sample was composed of by 494 participants in adolescent age. The re-contact success rate was 38.5%.
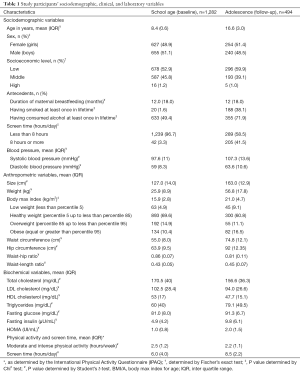
Most of the parents/guardians of the study population reported they belonged to middle and low socioeconomic levels (Table 1). With regards to the nutritional status in school age, there was 20% reported as overweight and 6.2% obesity, whereas in the adolescent stage the overweight decreased (14.9% vs. 11.1%) and obesity increased (10.5 vs. 16.5%) (Table 1).
Full table
Concerning the risk factors related to behaviors in the school age, 93.2% reported doing moderate physical activity and 37.4% did intense activity. By the time of follow-up in adolescence, these proportions decreased to 91.4% and 21.7% respectively.
Moreover, 3.3% of children in school age versus 41.5% of participants in adolescent ages reported spending more than 8 hours per week in front of screens (television sets, videogames, and tablets).
With regards to biochemical assessment of lipids, total cholesterol and LDL cholesterol, elevated values were found on the baseline, which remained stable at the time of follow-up.
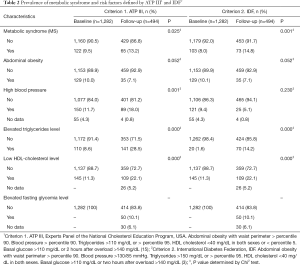
In relation to the MS, prevalence on the baseline as defined by ATP III was greater than the one established by the IDF, with a statistically significant difference (P=0.037). This prevalence increased 3.7% by using the ATP III criteria and 6.8% by using the IDF at the time of follow-up (Table 2).
Full table
When comparing the prevalence of the MS in boys and girls in school age against the prevalence in a sub sample of this population during the adolescent stage, significant differences were found using both the ATP III criteria (P=0.025) and the IDF ones (P=0.001), being greater in the adolescence stage.
Similarly, with regards to each one of the risk factors that make up the definition of MS by means of the criteria used, the elevated triglycerides level and low HDL cholesterol level had significant differences between the baseline and the follow-up using both criteria. In the case of abdominal obesity those differences were not found, while prevalence of high blood pressure was greater in the adolescent stage compared against the school age (P=0.001) (Table 2).
Discussion
This study shows the prevalence of MS among children and adolescents who are part of the first population-based cohort in Colombia for the last ten years in order to study of cardiovascular risk factors in early stages of live (13). When comparing the prevalence of MS among children and adolescents, an increase of this outcome was observed. Additionally, this prevalence is greater when the IDF criteria are employed compared against the one resulting from the ATP III criteria.
In a recent publication for the Americas, 23 articles were mentioned including three publications for Colombia in populations with age arrays similar to the ones described in this paper, in a period encompassing from 2008 to 2016. Studies included in this publication reported that there is variability in the MS prevalence, being less than 6% in Argentina, Colombia, Guatemala, Mexico, and Paraguay whereas in Brazil, Canada, and Venezuela it was greater than 12.0% (16). This study evinced a prevalence of 9.5% for MS in 2006, value which is higher than the ones reported in three studies conducted in Colombian population; two in the city of Medellin (5.1% and 3.1%) (17,18), and another in the city of Cali (8.7%) (19) by using the ATP III diagnosis criteria (15). Similarly, the population analyzed in this study reported 10 years after the baseline measurement in school age, an approximate increase of 4% points (13.21%) in the presence of MS taking into account the ATP III criteria.
It is important to note the difference in the cut points used by both diagnosis criteria employed in this study, in order to define the MS. This variability might explain the greater prevalence of the disease. For instance, for the case of triglycerides, the IDF criterion considers them elevated with a figure exceeding 150 mg/dL (3), whereas for the ATP III criterion the limit number is 110 mg/dL (15). These differences in the cut points have implications on the estimation of the prevalence of the disease, especially when it is considered that hypertriglyceridemia is the component of the MS that appears with most frequency in an isolated manner. For the study population, 8.6% in school age and 28.5% in adolescent age had a concentration of triglycerides >110 mg/dL, prevalence that decreased 66% in average for the two times of evaluation when considering the cut point of 150 mg/dL. According to Agudelo Ochoa et al. (17), who evaluate MS as per ATP III, the outcomes for elevated triglycerides are similar to the ones found in this study, being the follow-up participants the ones that most approach that prevalence. Prevalence found in this study is almost three times lower compared to the one found by Múnera et al. (40.9%) (18). On the other hand, by considering the IDF criteria (3) there is evidence that the prevalence of this factor for the baseline is very low since, according to their cut points, they only apply for population above 10 years of age. However, the findings of this study are similar to what was reported in the Chilean population (8.7%) (20), unlike the reported in Canada (4%) (21), which is three times below, and in Mexico (24.4%) (22) which doubles the value reported herein.
With regards to prevalence of the other factors (abdominal obesity, high blood pressure, low HDL cholesterol level, elevated fasting glycemia level) which comprise MS as per ATP III and the IDF are very variable and they fluctuate from 10% to 30% approximately. Reports in the literature show a wider variability ranging from 9% to 70% (16). It is worth to highlight that this paper and the review conducted by Pierlot et al. (16) evince that there is still a lack of consensus among expert researchers and institutions who are responsible for setting up the detection criteria.
On the other hand, one of the most relevant findings in our study is the fact that boys and girls (of school age) have an average screen time of 6.0 hours per day, number that increased in the follow-up stage to 8.5 hours per day. This average represents more than four times what is recommended by the American Academy of Pediatrics (23) and might represent a negative impact in terms of percent body fat, abdominal circumference, waist-length ratio, and BMI.
The main strengths of this study are based on the methodological soundness of both the original cohort and the follow-up data, the commitment of its researchers and the national and international institutions which support them. This constitutes the ideal starting point to analyze the influence that ethnic and environmental variants may have on the behavior of cardiovascular risk factors at early ages.
The main limitation of this paper was the low percentage of success in re-contacting, which is explained by high mobility of the participants of the original cohort to other cities or neighborhoods, which hindered their location.
Conclusions
This study shows that the prevalence of the MS in the population studied increased 4%, from 9.5% in children of school age to 13.1% in adolescents. This increase in the MS deserves special attention from early ages of life with the aim of decreasing metabolic, cardiovascular, and cerebrovascular damages in adulthood. An early intervention for the promotion of healthy lifestyles including increase in physical activity, decrease of practices demanding inactivity time such as the use of screens (tablets, mobile devices, computers, etc.), balanced diets with nutritional content according to the age, adaptation of public spaces for outdoor recreation, and improvement of access to health services that allow early diagnosis and timely treatment.
Supplementary
- Sociodemographic variables: sex, age, socioeconomic level.
- Anthropometric variables: weight, size, body max index, waist circumference, hip circumference, waist-length index (WLI), waist-hip index (WHI).
- Behavior variables: alcohol and tobacco use, moderate and intense physical activity, screen time.
- Cardiometabolic variables considered: impaired blood pressure: systolic blood pressure (SBP) or diastolic blood pressure (DBP) ≥95 percentile for age, gender, and size measured in three or more opportunities. Pre-hypertension: SBP or DBP ≥90 percentile for age, gender, and size (the fourth report on the diagnosis, evaluation and treatment of high blood pressure in children and adolescents). Overweight or obesity: overweight: body mass index (BMI) for the age between percentiles from 85% to 95%. Obesity: ≥ percentile 95 BMI for the age (Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents. National Heart, Lung and Blood Institute, NHI. USA, 2012). Impaired fasting glucose: ≥100, diabetes: ≥126 mg/dL (Standards of Medical Care in Diabetes, ADA 2016). HOMA-IR: obtained from a mathematical model using the formula [(IF*GF)/22.5], where IF represents insulin levels at fasting in UL/L and GF represents fasting glucose levels. Dyslipidemia: *alteration in total cholesterol: <170 mg/dL, *alteration in LDL cholesterol: ≥130 mg/dL (Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents. National Heart, Lung and Blood Institute, NHI. USA, 2012).
Acknowledgments
Our gratitude to funding from Colciencias within the CARDIECOL program (Spanish acronym for knowledge and action to reduce the dimension of cardiovascular disease in Colombia) by means of contract 376-2011, and researchers from the projects “Prevalence of the metabolic syndrome in the population aged 6 to 10 residing in Bucaramanga during the term comprised from April 2006 and April 2007, SIMBA phase I”, and “Assembly of a cohort to evaluate the early presentation of cardiovascular and metabolic dysfunction in adolescents with Metabolic Syndrome, SIMBA phase II”, who facilitated the database in order to derive the information necessary to develop this paper.
Funding: Colciencias within the CARDIECOL program (Spanish acronym for knowledge and action to reduce the dimension of cardiovascular disease in Colombia) by means of contract 376-2011. Colciencias is the Administrative Department of Science, Technology and Innovation in Colombia. Promotes public policies to promote Science, Technology and Innovation (CT + I) in Colombia. The activities surrounding the fulfillment of its mission involve agreeing policies to promote the production of knowledge, build capacities for CT + I, and promote circulation and uses of them for the integral development of the country and the well-being of Colombians. This entity only finances through public convocation. Among the commitments that are created is the publication of results of the project financed in high impact journals. This entity has no interference in the design of the study and the collection, analysis and interpretation of the data and in the writing of the manuscript. Each researcher is autonomous.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the Committee for Ethics in Research of Fundación Cardiovascular de Colombia in act No. 396 of April 9, 2016. Consent was obtained from the parents or legal guardians of the participants by written. Additionally, all participants of school age gave their verbal assent and they signed an agreement (assent), and for the follow-up those teenagers of legal age (18 years) gave their written consent.
References
- Reaven GM. Role of insulin resistance in human disease. Banting lecture 1988. Diabetes 1988;37:1595-607. [Crossref] [PubMed]
- Day C. Metabolic syndrome, or What you will: definitions and epidemiology. Diab Vasc Dis Res 2007;4:32. [Crossref] [PubMed]
- Zimmet P, Alberti G, Kaufman F, et al. International Diabetes Federation Task Force on Epidemiology and Prevention of Diabetes: The metabolic syndrome in children and adolescents. Lancet 2007;369:2059-61. [Crossref] [PubMed]
- Reinehr T. Metabolic syndrome in children and adolescents: a critical approach considering the interaction between pubertal stage and insulin resistance. Curr Diab Rep 2016;16:8-17. [Crossref] [PubMed]
- Moreno LJ, Velasco-Benítez CA, Matallana AM. Síndrome Metabólico en niños y adolescents. Revista Gastrohnup 2014;16:121-31.
- Ewald DR, Haldeman LA. Risk factors in adolescent hypertension. Glob Pediatr Health 2016;3:2333794X15625159.
- Kim JY, Tfayli H, Michaliszyn SF, et al. Distinguishing characteristics of metabolically healthy versus metabolically unhealthy obese adolescent girls with polycystic ovary syndrome. Fertil. Steril 2016;105:1603-11. [Crossref] [PubMed]
- Bouzas IC, Cader SA, Leão L, Kuschnir MC, et al. Menstrual cycle alterations during adolescence: early expression of metabolic syndrome and polycystic ovary syndrome. J Pediatr Adolesc Gynecol 2014;27:335-41. [Crossref] [PubMed]
- McPherson NO, Lane M. Male obesity and subfertility, is it really about increased adiposity? Asian J Androl 2015;17:450-8. [PubMed]
- Lawrence RS, Gootman JA; Sim LJ. Adolescent Health Services: Missing Opportunities. National Academies Press, Washington, DC 2009. National Research Council (U.S.) 2009.
- PAHO. Adolescents & non-communicable diseases. Panamerican Health Organization, Washington, DC 2011. Available online: www.paho.org/ncds
- World Health Organization. The world health report 2002: Reducing risks, promoting healthy life World Health Organization, Geneva, 2002.
- Villa-Roel C, Buitrago A, Rodríguez D, et al. Prevalence of metabolic syndrome in scholars from Bucaramanga, Colombia: a population-based study. Study protocol. BMC Pediatrics 2009;9:28. [Crossref] [PubMed]
- Gamboa Delgado EM, Domínguez-Urrego CL, Quintero-Lesmes DC. Waist-to-height ratio and its relation with cardiometabolic risk factors in children from Bucaramanga, Colombia. Nutrición Hospitalaria 2017;34:1338-44. [PubMed]
- Executive Summary of the Third Report of the National Cholesterol Education Program NCEP Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults Adult Treatment Panel III. JAMA 2001;285:2486-97. [Crossref] [PubMed]
- Pierlot R, Cuevas-Romero E, Rodríguez-Antolín J, et al. Prevalencia de síndrome metabólico en niños y adolescentes de América. Disponible en: ). TIP Revista Especializada en Ciencias Químico-Biológicas 2017;20:40-9.http://creativecommons.org/licenses/by-nc-nd/4.0/
- Agudelo Ochoa GM, Arias Arteaga R. Prevalencia del síndrome metabólico en niños y adolescentes escolarizados del área urbana de la ciudad de Medellín. Iatreia 2008;21:260-70.
- Múnera NE, Uscátegui R, Parra B, et al. Factores de riesgo ambientales y componentes del síndrome metabólico en adolescentes con exceso de peso. Biomédica 2012;32:77-91. [PubMed]
- Suarez-Ortegón MF, Aguilar-de Plata C. Prevalence of metabolic syndrome in children aged 5-9 years from southwest Colombia: a cross-sectional study. World. J. Pediatr 2016;12:477-83. [Crossref] [PubMed]
- Burrows R, Correa-Burrows P, Reyes M, et al. High cardiometabolic risk in healthy Chilean adolescents: associations with anthropometric, biological and lifestyle factors. Public Health Nutr 2016;19:486-93. [Crossref] [PubMed]
- Wahi G, Zorzi A, Macnab A, et al. Prevalence of type 2 diabetes, obesity and the metabolic syndrome among Canadian First Nations children in a remote Pacific coast community. Paediatr Child Health 2009;14:79-83. [PubMed]
- Cárdenas-Villarreal VM, López-Alvarenga JC, Bastarrachea RA, et al. Prevalencia del síndrome metabólico y sus componentes en adolescentes de la Ciudad de Monterrey, Nuevo León. Arch Cardiol Mex 2010;80:19-26.
- AAP Council on communications and media. Media and Young Minds. Pediatrics 2016;138:e20162591. [Crossref]


